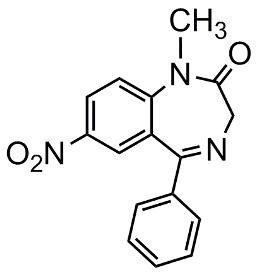
Trade names Erimin ATC code N05 CAS ID 2011-67-8 | Pregnancycategory X Molar mass 295.3 g/mol Bioavailability 95% | |
AHFS/Drugs.com International Drug Names Routes ofadministration OralAddiction liability: High Legal status AU: S4 (Prescription only)CA: Schedule IVDE: Anlage III (Prescription only)US: Schedule IVUN: Psychotropic Schedule IV | ||
Elextrolex tribaltek nimetazepam overdose mix
Nimetazepam (marketed under brand name Erimin and Lavol) is an intermediate-acting hypnotic drug which is a benzodiazepine derivative. It was first synthesized by a team at Hoffmann-La Roche in 1962. It possesses hypnotic, anxiolytic, sedative, and skeletal muscle relaxant properties. Nimetazepam is also an anticonvulsant. It is sold in 5 mg tablets known as Erimin and Lavol. It is generally prescribed for the short-term treatment of severe insomnia in patients who have difficulty falling asleep or maintaining sleep. The sole global manufacturer of Nimetazepam (Sumitomo Japan) has ceased manufacturing Erimin since early November 2015. Patients being prescribed Erimin are being switched to other hypnotics, e.g. etizolam, Flunitrazepam, etc.
Contents
- Elextrolex tribaltek nimetazepam overdose mix
- Pharmacokinetics
- Recreational use
- Legal status
- Toxicity
- References

Pharmacokinetics

Taken orally, nimetazepam has very good Bioavailability with nearly 100% being absorbed from the gut. It is among the most rapidly absorbed and quickest acting oral Benzodiazepines, and hypnotic effects are typically felt within 15–30 minutes after oral ingestion. The blood level decline of the parent drug was biphasic with the short half-life ranging from 0.5–0.7 hours and the terminal half-life from 8 to 26.5 hours (mean 17.25 hours). It is the N-methylated analogue of nitrazepam (Mogadon, Alodorm), to which it is partially metabolized. nitrazepam has a long Elimination half-life, so effects of repeated dosage tend to be cumulative.
Recreational use

Nimetazepam has a particular reputation in the Malay Peninsula for recreational use, at RM 25 per tab, and is particularly popular among persons addicted to amphetamines or opioids.
Legal status
Nimetazepam is currently a Schedule IV drug under the international Convention on Psychotropic Substances of 1971.
In Singapore, nimetazepam is a class A drug under the Misuse of Drugs Act.
In Hong Kong, nimetazepam is regulated under Schedule 1 of Hong Kong's Chapter 134 Dangerous Drugs Ordinance. Nimetazepam can only be used legally by health professionals and for university research purposes. The substance can be given by pharmacists under a prescription. Anyone who supplies the substance without prescription can be fined $10000 (HKD). The penalty for trafficking or manufacturing the substance is a $5,000,000 (HKD) fine and life imprisonment. Possession of the substance for consumption without license from the Department of Health is illegal with a $1,000,000 (HKD) fine and/or 7 years of jail time.
In Victoria Australia, nimetazepam is regulated under Schedule 11 of "Drugs, Poisons and Controlled substances act 1981". It is deemed to fall under the category of "7-NITRO-1,4-BENZODIAZEPINES not included elsewhere in this Part". .
Toxicity

In a rat study Nimetazepam showed greater damage to the fetus, as did nitrazepam when compared against other benzodiazepines, all at a dosage of 100 mg/kg. diazepam however showed relatively weak fetal toxicities. The same fetotoxicity of nitrazepam could not be observed in mice and is likely due to the particular Metabolism of the drug in the rat.
