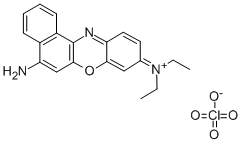
Formula C20H20ClN3O | Molar mass 353.845 g/mol | |
 | ||
Nile blue (or Nile blue A) is a stain used in biology and histology. It may be used with live or fixed cells, and imparts a blue colour to cell nuclei.
Contents
- Chemical and physical properties
- Nile blue staining
- Workflow
- Results
- Example Detection of poly hydroxybutyrate granules PHB
- Nile blue in DNA Electrophoresis
- Nile blue in oncology
- Synthesis
- References
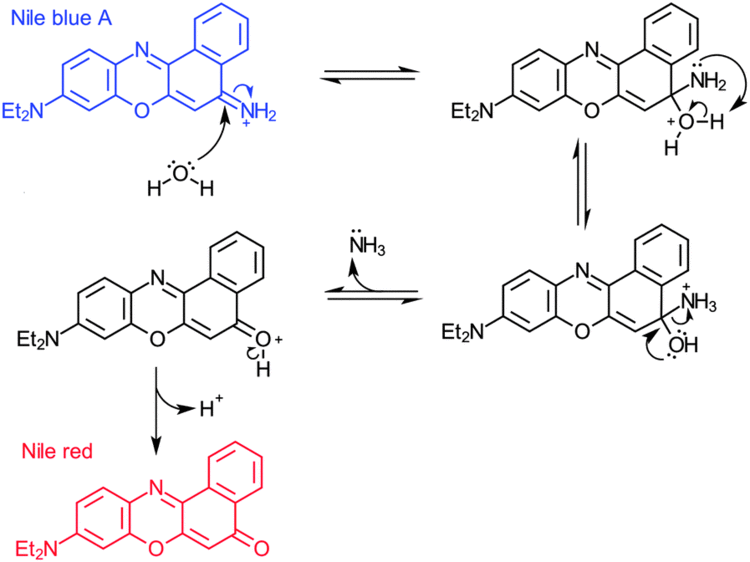
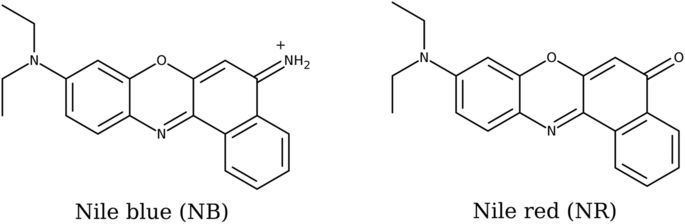
It may also be used in conjunction with fluorescence microscopy to stain for the presence of polyhydroxybutyrate granules in prokaryotic or eukaryotic cells. Boiling a solution of Nile blue with sulfuric acid produces Nile red (Nile blue oxazone).
Chemical and physical properties
Nile blue is a fluorescent dye. The fluorescence shows especially in nonpolar solvents with a high quantum yield.
The absorption and emission maxima of Nile blue are strongly dependent on pH and the solvents used:
The duration of Nile blue fluorescence in ethanol was measured as 1.42 ns. This is shorter than the corresponding value of Nile red with 3.65 ns. The fluorescence duration is independent on dilution in the range 10−3–10−8 mol/L.
Nile blue staining
Nile blue is used for histological staining of biological preparations. It highlights the distinction between neutral lipids (triglycerides, cholesteryl esters, steroids) which are stained pink and acids (fatty acids, chromolipids, phospholipids) which are stained blue.
The Nile blue staining, according to Kleeberg, uses the following chemicals:
Workflow

The sample or frozen sections is/are fixated in formaldehyde, then immersed for 20 minutes in the Nile blue solution and rinsed with water. For better differentiation, it is dipped in 1% acetic acid for 10–20 minutes until the colors are pure. This might take only 1–2 minutes. Then the sample is thoroughly rinsed in water (for one to two hours). Afterwards, the stained specimen is taken on a microscope slide and excess water is removed. The sample can be embedded in glycerol or glycerol gelatin.
Results
Unsaturated glycerides are pink, nuclei and elastins(?) dark, fatty acids and various fatty substances and fat mixtures are purple blue.
Example: Detection of poly-β-hydroxybutyrate granules (PHB)
The PHB granules in the cells of Pseudomonas solanacearum can be visualized by Nile blue A staining. The PHB granules in the stained smears are observed with an epifluorescence microscope under oil immersion, at a 1000 times magnification; under 450 nm excitation wavelength they show a strong orange fluorescence.
Nile blue in DNA Electrophoresis
Nile blue is also apparently used in a variety of commercial DNA staining formulations used for DNA electrophoresis. As it does not require UV trans-illumination in order to be visualised in an agarose gel as with ethidium bromide, it can be used to observe DNA as it is separated and also as a dye to aid in gel-extraction of DNA fragments without incurring damage by UV-irradiation.
Nile blue in oncology
Derivatives of Nile blue are potential photosensitizers in photodynamic therapy of malignant tumors. These dyes aggregate in the tumor cells, especially in the lipid membranes, and/or are sequestered and concentrated in subcellular organelles.
With the Nile blue derivative N-Ethyl-Nile Blue (EtNBA), normal and premalignant tissues in animal experiments can be distinguished by fluorescence spectroscopy in fluorescence imaging. EtNBA shows no phototoxic effects.
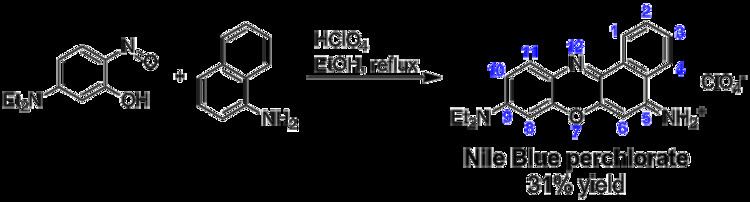
Synthesis
Nile Blue and related naphthoxazinium dyes can be prepared by acid-catalyzed condensation of either 5-(dialkylamino)-2-nitrosophenols with 1-naphthylamine, 3-(dialkylamino)phenols with N-alkylated 4-nitroso-1-naphtylamines, or N,N-dialkyl-1,4-phenylenediamines with 4-(dialkylamino)-1,2-naphthoquinones. Alternatively, the product of an acid-catalyzed condensation of 4-nitroso-N,N-dialkylaniline with 2-naphthol (a salt of 9-(dialkylamino)benzo[a]phenoxazin-7-ium) can be oxidized in the presence of an amine, installing a second amino substituent in 5-position of the benzo[a]phenoxazinium system. The following scheme illustrates the first of these four approaches, leading to Nile Blue perchlorate:
