 | ||
lactams mechanisms of action and resistance
A lactam is a cyclic amide. The term is a portmanteau of the words lactone + amide.
Contents
- lactams mechanisms of action and resistance
- Beta lactam antibiotics carboxylic acids and derivatives organic chemistry khan academy
- Nomenclature
- Synthesis
- Tautomerization to Lactim
- Reactions
- References
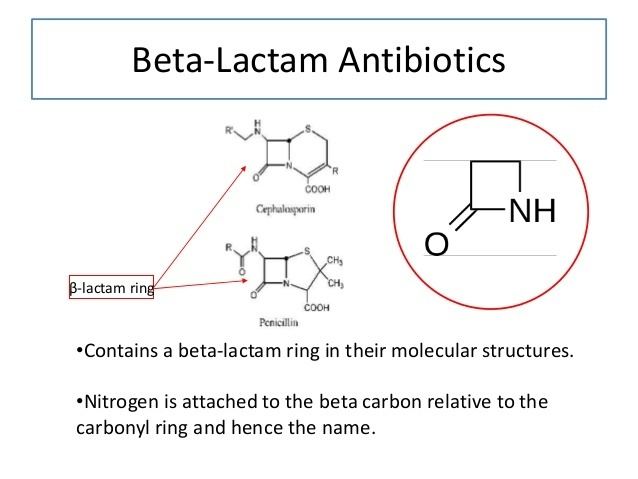
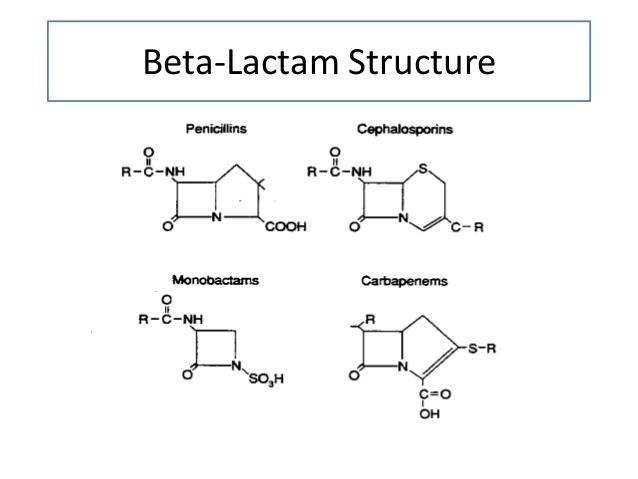
Beta lactam antibiotics carboxylic acids and derivatives organic chemistry khan academy
Nomenclature
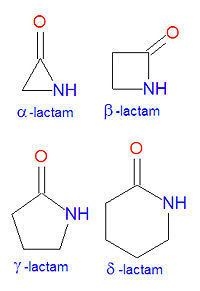
Greek prefixes in alphabetical order indicate ring size:
This ring-size nomenclature stems from the fact that a hydrolyzed α-Lactam leads to an α-amino acid and a β-Lactam to a β-amino acid, etc.
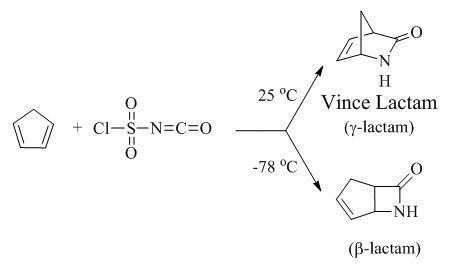
Synthesis
General synthetic methods exist for the organic synthesis of lactams.
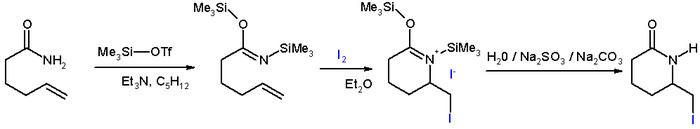
Tautomerization to Lactim
Lactim is a cyclic carboximidic acid compound characterized by an endocyclic carbon-nitrogen double bond. It is formed when lactam undergoes tautomerization.
Reactions

References
Lactam Wikipedia(Text) CC BY-SA
