In biochemistry and pharmacology, the binding of a ligand to a macromolecule is often enhanced if there are already other ligands present on the same macromolecule (this is known as cooperative binding). The Hill equation, which was originally formulated by Archibald Hill in 1910 to describe the sigmoidal O2 binding curve of haemoglobin, is used to describe the fraction of a macromolecule saturated by ligand as a function of the ligand concentration. The equation is useful for determining the degree of cooperativity of the ligand(s) binding to the enzyme or receptor. The Hill coefficient provides a way to quantify the degree of interaction between ligand binding sites.
The Hill equation is formally equivalent to the Langmuir isotherm, and as a result, it is sometimes referred to as the Hill-Langmuir equation.
The Hill equation is commonly expressed in the following ways:
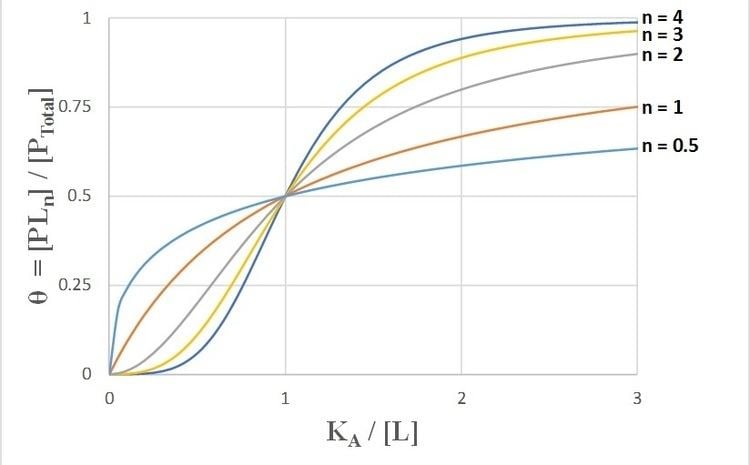
θ = [ L ] n K d + [ L ] n = [ L ] n ( K A ) n + [ L ] n = 1 ( K A [ L ] ) n + 1 ,
The equation's terms are defined as follows:
θ - Fraction of the receptor protein concentration that is bound to ligand. [ L ] - Free (unbound) ligand concentration K d - Apparent dissociation constant derived from the law of mass action (the equilibrium constant for dissociation), which is equal to the ratio of the dissociation rate of the ligand-receptor complex to its association rate ( K d = k d k a ). K A - The ligand concentration producing half occupation (ligand concentration occupying half of the binding sites). Because K A is defined so that ( K A ) n = K d = k d k a , this is also known as the microscopic dissociation constant. In recent literature, this constant is sometimes referred to as K D . n - The Hill coefficient, describing cooperativity (or possibly other biochemical properties, depending on the context in which the Hill equation is being used)When appropriate, the value of the Hill coefficient describes the cooperativity of ligand binding in the following way:
n > 1 - Positively cooperative binding: Once one ligand molecule is bound to the enzyme, its affinity for other ligand molecules increases. For example, the Hill coefficient of oxygen binding to haemoglobin (an example of positive cooperativity) falls within the range of 1.7-3.2. n < 1 - Negatively cooperative binding: Once one ligand molecule is bound to the enzyme, its affinity for other ligand molecules decreases. n = 1 - Noncooperative (completely independent) binding: The affinity of the enzyme for a ligand molecule is not dependent on whether or not other ligand molecules are already bound. When n=1, we obtain a model that can be modeled by Michaelis–Menten kinetics, in which K D = K A = K M , the Michaelis-Menten constant.Taking the reciprocal of both sides of the Hill equation, rearranging, and inverting again yields: θ 1 − θ = [ L ] n K d = [ L ] n ( K A ) n .
Taking the logarithm of both sides of the equation leads to an alternative formulation of the Hill equation:
log ( θ 1 − θ ) = n log [ L ] − log K d = n log [ L ] − n log K A .
This last form of the Hill equation is advantageous because a plot of log ( θ 1 − θ ) versus log [ L ] yields a linear plot, which is called a Hill plot. Because the slope of a Hill plot is equal to the Hill Coefficient for the biochemical interaction, the slope is denoted by n H . A slope greater than one thus indicates positively cooperative binding between the receptor and the ligand, while a slope less than one indicates negatives cooperative binding.
Consider a protein ( P ), such as Haemoglobin or a protein receptor, with n binding sites for ligands ( L ). The binding of the ligands to the protein can be represented by the chemical equilibrium expression:
P + n L ⇌ k d k a P L n ,
where k a (forward rate, or the rate of association of the protein-ligand complex) and k d (reverse rate, or the complex's rate of dissociation) are the reaction rate constants for the association of the ligands to the protein and their dissociation from the protein, respectively. From the law of mass action, the apparent dissociation constant K d , an equilibrium constant, is given by:
K d = k d k a = [ P ] [ L ] n [ P L n ] .
At the same time, θ , the ratio of the concentration of occupied receptor to total receptor concentration, is given by:
θ = O c c u p i e d R e c e p t o r T o t a l R e c e p t o r = [ P L n ] [ P ] + [ P L n ] .
By using the expression obtained earlier for the dissociation constant, we can replace [ P L n ] with [ P ] [ L ] n K d to yield a simplified expression for θ :
θ = ( [ P ] [ L ] n K d ) [ P ] + ( [ P ] [ L ] n K d ) = [ P ] [ L ] n K d [ P ] + [ P ] [ L ] n = [ L ] n K d + [ L ] n ,
which is a common formulation of the Hill equation.
Assuming that the protein receptor was initially completely free (unbound) at a concentration [ P 0 ] , then at any time, [ P ] + [ P L n ] = [ P 0 ] and θ = [ P L n ] [ P 0 ] . Consequently, the Hill Equation is also commonly written as an expression for the concentration [ P L n ] of bound protein:
[ P L n ] = [ P 0 ] ⋅ [ L ] n K d + [ L ] n .
Note that all of these formulations assume that the protein has n sites to which ligands can bind. In practice, however, the Hill Coefficient n rarely provides an accurate approximation of the number of ligand binding sites on a protein. Consequently, it has been observed that the Hill Coefficient should instead be interpreted as an "interaction coefficient" describing the cooperativity among ligand binding sites.
The Hill equation can be applied in modeling the rate at which a gene product is produced when its parent gene is being regulated by transcription factors (e.g., activators and/or repressors). Doing so is appropriate when a gene is regulated by multiple binding sites for transcription factors, in which case the transcription factors may bind the DNA in a cooperative fashion.
If the production of protein from gene X is up-regulated (activated) by a transcription factor Y , then the rate of production of protein X can be modeled as a differential equation in terms of the concentration of activated Y protein:
d [ X p r o d u c e d ] d t = k ⋅ [ Y a c t i v e ] n ( K A ) n + [ Y a c t i v e ] n ,
where k is the maximal transcription rate of gene X .
Likewise, if the production of protein from gene Y is down-regulated (repressed) by a transcription factor Z , then the rate of production of protein Y can be modeled as a differential equation in terms of the concentration of activated Z protein:
d [ Y p r o d u c e d ] d t = k 1 + ( [ Y a c t i v e ] K A ) n .
Because of its assumption that ligand molecules bind to a receptor simultaneously, the Hill equation has been criticized as a physically unrealistic model. Moreover, the Hill coefficient should not be considered a reliable approximation of the number of cooperative ligand binding sites on a receptor except when the binding of the first and subsequent ligands results in extreme positive cooperativity.
Unlike more complex models, the relatively simple Hill equation provides little insight into underlying physiological mechanisms of protein-ligand interactions. This simplicity, however, is what makes the Hill equation a useful empirical model, since its use requires little a priori knowledge about the properties of either the protein or ligand being studied. Nevertheless, other, more complex models of cooperative binding have been proposed. For more information and examples of such models, see Cooperative binding.