Formula C7H8O2 Boiling point 205 °C | Density 1.11 g/cm³ Molar mass 124.14 g/mol | |
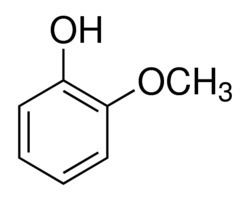 | ||
Guaiacol is a naturally occurring organic compound with the formula C6H4(OH)(OCH3), first isolated by Otto Unverdorben in 1826. Although it is biosynthesized by a variety of organisms, this yellowish aromatic oil is usually derived from guaiacum or wood creosote. Samples darken upon exposure to air and light. Guaiacol is present in wood smoke, resulting from the pyrolysis of lignin. The compound contributes to the flavor of many compounds, e.g., roasted coffee.
Contents
Preparation
Guaiacol is produced by methylation of catechol, e.g., using potash and dimethyl sulfate:
C6H4(OH)2 + (CH3O)2SO2 → C6H4(OH)(OCH3) + HO(CH3O)SO2Laboratory methods
Guaiacol can be prepared by diverse routes in the laboratory. 2-Aminoanisole, derived in two steps from anisole, can be hydrolyzed via its diazonium derivative. Guaiacol can be synthesized by the dimethylation of catechol followed by selective mono-demethylation.
C6H4(OCH3)2 + C2H5SNa → C6H4(OCH3)(ONa) + C2H5SCH3Uses and chemical reactions
Guaiacol is a precursor to various flavorants, such as eugenol and vanillin. An estimated 85% of the worlds supply of vanillin comes from guaiacol. The route entails condensation of glyoxylic acid with guaiacol to give mandelic acid, which is oxidized to produce a phenylglyoxylic acid. This acid undergoes a decarboxylation to afford vanillin.
In basic solution, it is readily oxidized, giving yellowish brown and absorbs light maximally at about 470 nm.
Its derivatives are used medicinally as an expectorant, antiseptic, and local anesthetic.
Related compounds
Guaiacol carbonate is known as duotal, the phosphate as phosphatol, the phosphite as guaiaco-phosphal (phosphotal is a mixture of the phosphites of creosote phenols). The valerianic ester of guaiacol is known as geosote, the benzoic as benzosol, the salicylic as guaiacolsalol, while the glycerin ether is the drug guaifenesin. The related derivative, dimethoxybenzene or veratrole, is also useful. In preparation of food by smoking, guaiacol is the main chemical responsible for the smoky taste, whereas syringol is responsible for the smoky aroma.
Safety
Methoxyphenols are potential biomarkers of biomass smoke exposure, e.g., from inhalation of woodsmoke. Dietary sources of methoxyphenols overwhelm the contribution from inhalational exposures to woodsmoke.
Locust pheromone
Guaiacol is produced in the gut of desert locusts, Schistocerca gregaria, by the breakdown of plant material. This process is undertaken by the gut bacterium Pantoea agglomeransa (Enterobacter). It is one of the main components of the pheromones that cause locust swarming.
