AHFS/Drugs.com Monograph CAS ID 126-07-8 | MedlinePlus a682295 Molar mass 352.766 g/mol | |
 | ||
Trade names Gris-peg, Grifulvin V, others Pregnancycategory AU: B3US: C (Risk not ruled out) | ||
Griseofulvin is antifungal medication used to treat a number of types of dermatophytoses (ringworm). This includes fungal infections of the nails and skin when antifungal creams have not worked. It is taken by mouth.
Contents

Common side effects include allergic reactions, nausea, diarrhea, headache, trouble sleeping, and feeling tired. It is not recommended in people with liver failure or porphyria. Use during or in the months before pregnancy may result in harm to the baby. Griseofulvin works by interfering with fungal mitosis.
Griseofulvin was discovered in 1939 from a type of Penicillium mold. It is on the World Health Organization's List of Essential Medicines, the most effective and safe medicines needed in a health system. The wholesale cost in the developing world is about 0.05 to 0.18 USD per day. In the United States a course of treatment costs 100 to 200 USD.
Medical uses
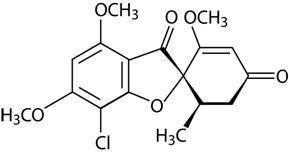
Griseofulvin is used orally only for dermatophytosis. It is ineffective topically. It is reserved for cases with nail, hair, or large body surface involvement.
Side effects
Known side effects of griseofulvin include:
Mechanism
The drug binds to tubulin, interfering with microtubule function, thus inhibiting mitosis. It binds to keratin in keratin precursor cells and makes them resistant to fungal infections. The drug reaches its site of action only when hair or skin is replaced by the keratin-griseofulvin complex. Griseofulvin then enters the dermatophyte through energy-dependent transport processes and bind to fungal microtubules. This alters the processing for mitosis and also underlying information for deposition of fungal cell walls.
Biosynthetic process
It is produced industrially by fermenting the fungus Penicillium patulum.
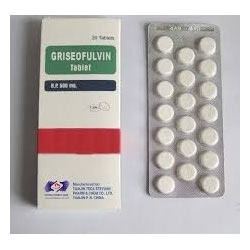
The first step in the biosynthesis of griseofulvin by P. griseofulvin is the synthesis of the 14-carbon poly-β-keto chain by a type I iterative polyketide synthase (PKS) via iterative addition of 6 malonyl-CoA to an acyl-CoA starter unit. The 14-carbon poly-β-keto chain undergoes cyclization/aromatization, using cyclase/aromatase, respectively, through a Claisen and aldol condensation to form the benzophenone intermediate. The benzophenone intermediate is then methylated via S-adenosyl methionine (SAM) twice to yield griseophenone C. The griseophenone C is then halogenated at the activated site ortho to the phenol group on the left aromatic ring to form griseophenone B. The halogenated species then undergoes a single phenolic oxidation in both rings forming the two oxygen diradical species. The right oxygen radical shifts alpha to the carbonyl via resonance allowing for a stereospecific radical coupling by the oxygen radical on the left ring forming a tetrahydrofuranone species. The newly formed grisan skeleton with a spiro center is then O-methylated by SAM to generate dehydrogriseofulvin. Ultimately, a stereoselective reduction of the olefin on dehydrogriseofulvin by NADPH affords griseofulvin.
Brand names