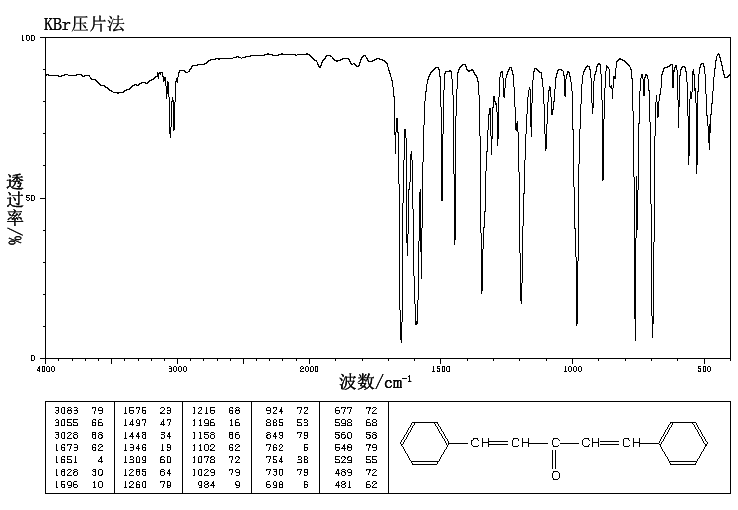
Molar mass 234.29 g/mol Appearance Yellow solid | Formula C17H14O | |
 | ||
Making dibenzylideneacetone
Dibenzylideneacetone or dibenzalacetone, often abbreviated dba, is an organic compound with the formula C17H14O. It is a pale-yellow solid insoluble in water, but soluble in ethanol. Dibenzylideneacetone is used as a component in sunscreens and as a ligand in organometallic chemistry.
Contents

Preparation
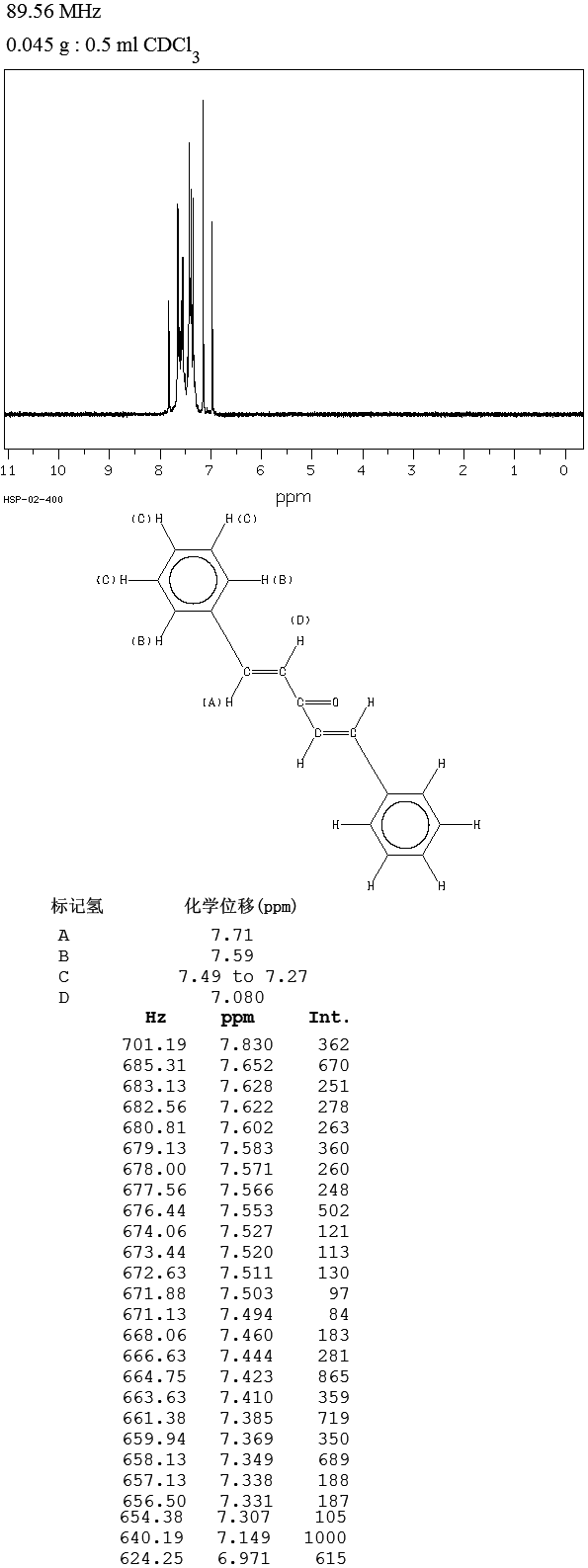
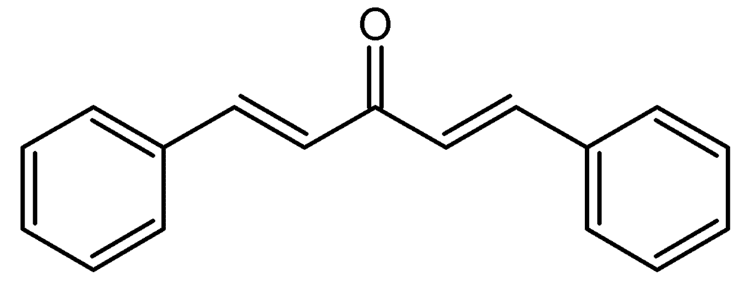
The compound can be prepared in the laboratory through condensation of benzaldehyde and acetone with sodium hydroxide in a water/ethanol medium with the exclusive formation of the trans, trans isomer (melting point 110–111 °C).
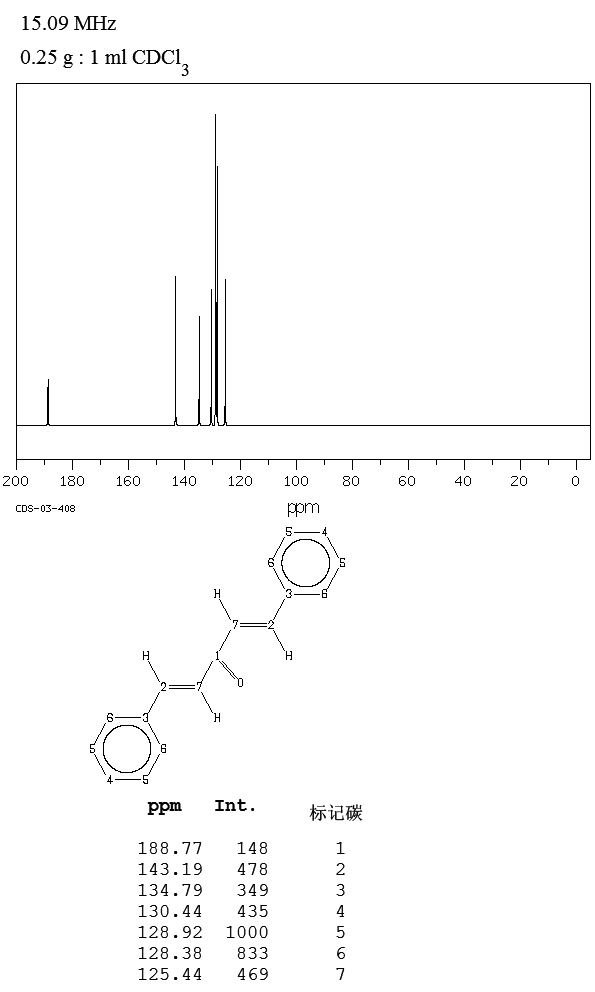
This reaction is frequently encountered in organic chemistry education as a laboratory procedure. The conversion proceeds via the intermediacy of benzylideneacetone.
Reactions and derivatives
Prolonged exposure to sunlight converts the compound in a [2+2] cycloaddition to a mixture of four cyclobutane isomers.
Dibenzylideneacetone is a component of the catalyst tris(dibenzylideneacetone)dipalladium(0). It is a labile ligand that is easily displaced by triphenylphosphine, hence it serves a useful entry point into palladium(0) chemistry.