 | ||
A condensation reaction is a chemical reaction in which two molecules or moieties, often functional groups, combine to form a larger molecule, together with the loss of a small molecule. Possible small molecules that are lost include water, acetic acid, hydrogen chloride, or methanol, but most commonly in biological reactions it is water.
Contents
When two separate molecules react, the condensation is termed intermolecular. A simple example is the condensation of two amino acids to form the peptide bond characteristic of proteins. This reaction example is the opposite of hydrolysis, which splits a chemical entity into two parts through the action of the polar water molecule, which itself splits into hydroxide and hydrogen ions. Hence energy is required to form chemical bonds via condensation.
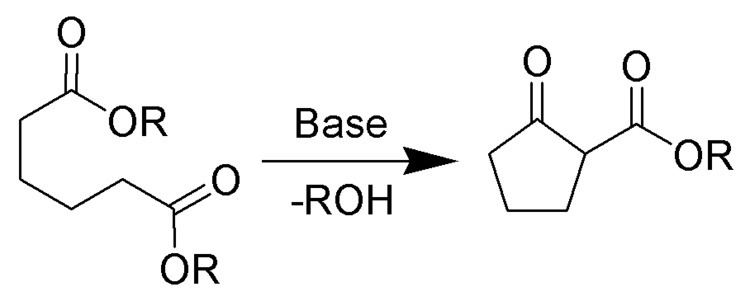
If the union is between atoms or groups of the same molecule, the intramolecular reaction is termed "intramolecular condensation", which in many cases leads to ring formation. An example is the Dieckmann condensation, in which the two ester groups of a single diester molecule react with each other to lose a small alcohol molecule and form a β-ketoester product.
Mechanism
Many condensation reactions follow a nucleophilic acyl substitution or an aldol condensation reaction mechanism. Other condensations, such as the acyloin condensation are triggered by radical or single electron transfer conditions.
Condensation polymerization
In condensation polymerization or "step-growth polymerization", multiple condensation reactions take place, joining monomers and monomer chains into long chains called polymers. It occurs for example in the synthesis of polyesters or nylons. It can be homopolymerization of a single monomer A-B with two different end groups that condense, or copolymerization of two co-monomers A-A and B-B.
Condensation polymerization releases multiple small molecules, in contrast to polyaddition reactions, which do not. In general, condensation polymers form more slowly than addition polymers, often requiring heat. They are generally lower in molecular weight. Monomers are consumed early in the reaction; the terminal functional groups remain active throughout; and short chains combine to form longer chains. A high conversion rate is required to achieve high molecular weights, per Carothers' equation.
Bifunctional monomers lead to linear chains, and therefore thermoplastic polymers, but, when the monomer functionality exceeds two, the product is a branched chain that may be a thermosetting polymer.
Applications
The reactions that form acid anhydrides from their constituent acids are typically condensation reactions.
Condensation polymerization produces many important polymers, for example: nylon, polyester, and other condensation polymers and various epoxies. It is also the basis for the laboratory formation of silicates and polyphosphates.
Many biological transformations are condensation reactions. Polypeptide synthesis, polyketide synthesis, terpene syntheses, phosphorylation, and glycosylation are a few examples. A large number of such reactions are used in synthetic organic chemistry. Other examples include:
Some reactions that are called "condensation reactions" for historical reasons, are actually addition reactions:
