Trade names Eumovate CAS Number 54063-32-0 | Routes ofadministration topical Molar mass 408.891 g/mol | |
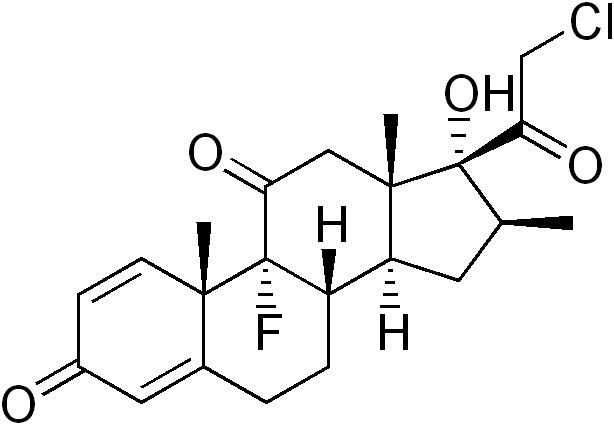 | ||
AHFS/Drugs.com Micromedex Detailed Consumer Information ATC code D07AB01 (WHO) S01BA09 (WHO) Synonyms (8S,9R,10S,13S,14S,16S,17R)-17-(2-Chloroacetyl)-9-fluoro-17-hydroxy-10,13,16-trimethyl-7,8,12,14,15,16-hexahydro-6H-cyclopenta[a]phenanthrene-3,11-dione | ||
Clobetasone (INN) is a corticosteroid used in dermatology, for treating such skin inflammation as seen in eczema, psoriasis and other forms of dermatitis, and ophthalmology. Topical clobetasone butyrate has shown minimal suppression of the Hypothalamic-pituitary-adrenal axis.
Contents
It is available as clobetasone butyrate under the brand names Eumosone or Eumovate both manufactured by GlaxoSmithKline.
Trimovate also contains Oxytetracycline, an antibiotic, and nystatin, an antifungal.
Uses
In dermatology, topical clobestasone butyrate helps to reduce the itchiness and erythema associated with eczema and dermatitis.
In ophthalmology, clobetasone butyrate 0.1% eye drops have been shown to be safe and effective in the treatment of dry eyes in Sjögren's Syndrome. Sjögren's Syndrome is an autoimmune disorder that affects the moisture producing glands of the body causing many symptoms including dry eyes. When compared to other corticosteroid eye drops; clobetasone butyrate showed only minimal rises in intraocular pressure. Increased pressure within the eye can lead to glaucoma.
Adverse effects
Side effects associated with clobetasone cream and ointment include: burning, irritation, itching, thinning of the skin, and changes in skin color.
