Formula HClO3 Molar mass 84.45914 g/mol Appearance colourless solution | IUPAC ID Chloric acid Density 1 g/cm³ | |
 | ||
Related compounds Thermodynamicdata Phase behavioursolid–liquid–gas Similar Chlorous acid, Perchloric acid, Nitric acid | ||
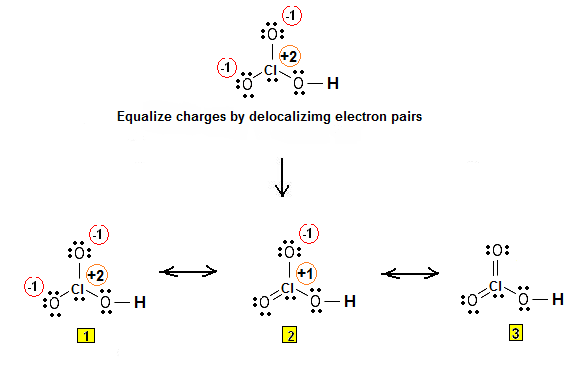
Chloric acid, HClO3, is an oxoacid of chlorine, and the formal precursor of chlorate salts. It is a strong acid (pKa ≈ −1) and oxidizing agent.
Contents
Properties
Chloric acid is thermodynamically unstable with respect to disproportionation.
Chloric acid is stable in cold aqueous solution up to a concentration of approximately 30%, and solution of up to 40% can be prepared by careful evaporation under reduced pressure. Above these concentrations, chloric acid solutions decompose to give a variety of products, for example:
8HClO3 → 4HClO4 + 2H2O + 2Cl2 + 3 O23HClO3 → HClO4 + H2O + 2 ClO2Hazards
Chloric acid is a powerful oxidizing agent. Most organics and flammables will deflagrate on contact.
Production
It can be prepared by the reaction of sulfuric acid with barium Chlorate, the insoluble barium sulfate being removed by precipitation:
Ba(ClO3)2 + H2SO4 → 2HClO3 + BaSO4Another method is the heating of hypochlorous acid, producing chloric acid and hydrogen chloride:
3HClO → HClO3 + 2 HCl