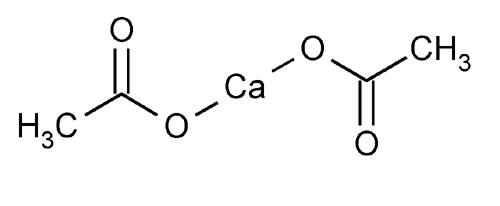
Abbreviations Ca(OAc)2 Density 1.6 g/cm³ | Melting point 160 °C Appearance White solid; hygroscopic | |
 | ||
Making calcium acetate from eggshells
Calcium acetate is a chemical compound which is a calcium salt of acetic acid. It has the formula Ca(C2H3O2)2. Its standard name is calcium acetate, while calcium ethanoate is the systematic name. An older name is acetate of lime. The anhydrous form is very hygroscopic; therefore the monohydrate (Ca(CH3COO)2•H2O) is the common form.
Contents
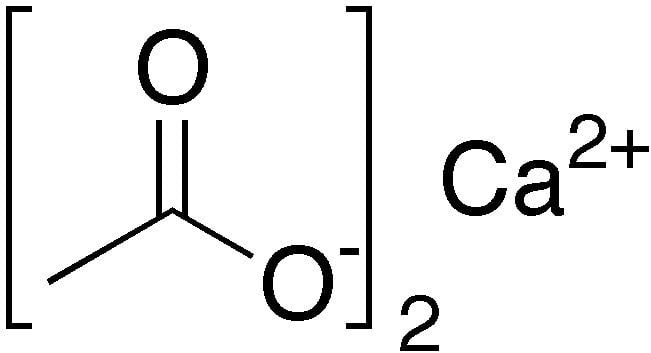
Make calcium acetate from tums
Production

Calcium acetate can be prepared by soaking calcium carbonate (found in eggshells, or in common carbonate rocks such as limestone or marble) or hydrated lime in vinegar:
CaCO3(s) + 2CH3COOH(aq) → Ca(CH3COO)2(aq) + H2O(l) + CO2(g)Ca(OH)2(s) + 2CH3COOH(aq) → Ca(CH3COO)2(aq) + 2H2O(l)Since both reagents would have been available pre-historically, the chemical would have been observable as crystals then.
Uses

References
Calcium acetate Wikipedia(Text) CC BY-SA
