 | ||
Borophene scientists create atomically thin boron
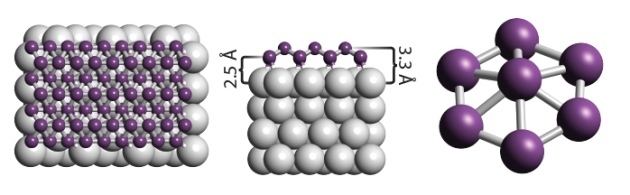
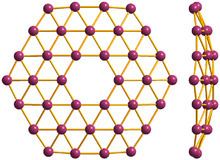
Borophene is a proposed crystalline allotrope of boron. One unit consists of 36 atoms arranged in an 2-dimensional sheet with a hexagonal hole in the middle. Another form made in 2015 is a buckled two dimensional sheet on silver.
Contents
- Borophene scientists create atomically thin boron
- Borophene joins 2d materials club scientists create atomically thin metallic boron borophene
- Theory
- History
- Borospherene
- References
Borophene joins 2d materials club scientists create atomically thin metallic boron borophene
Theory
Computational studies suggested that extended borophene sheets with partially filled hexagonal holes are stable. Global minimum searches for B−
36 lead to a quasiplanar structure with a central hexagonal hole. Borophene is predicted to be fully metallic.
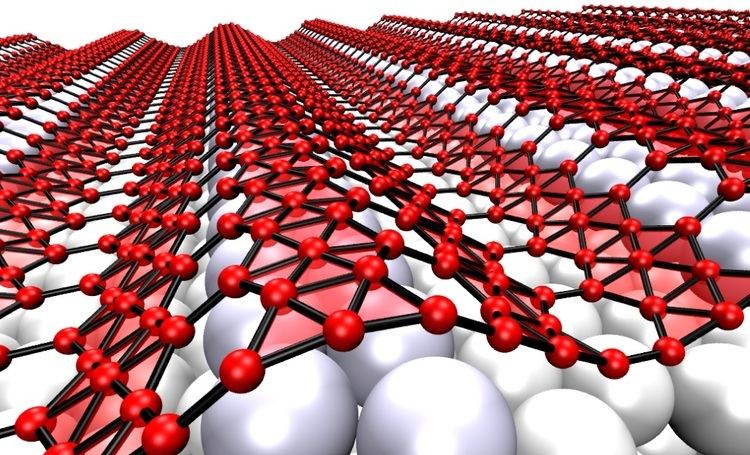
Borophene is analogous to graphene in that it is expected to form extended sheets. The latter is a semi-metal, implying that borophene may be a better conductor. The boron-boron bond is also nearly as strong as graphene’s carbon-carbon bond. At the atomic-cluster scale, pure boron forms simple planar molecules and cage-like fullerenes.
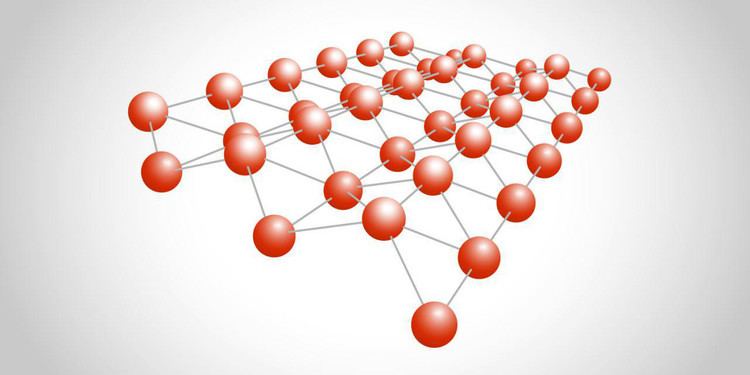
Boron is adjacent to carbon in the periodic table and has similar valence orbitals. Unlike carbon, boron cannot form a honeycomb hexagonal framework (like graphene) because of its electron deficiency.
History
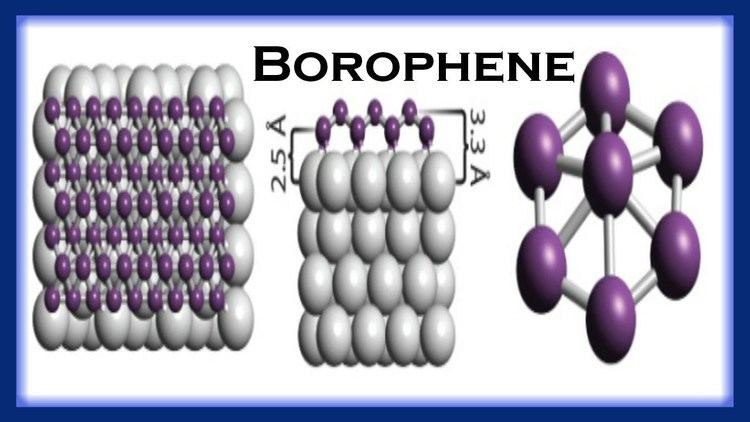
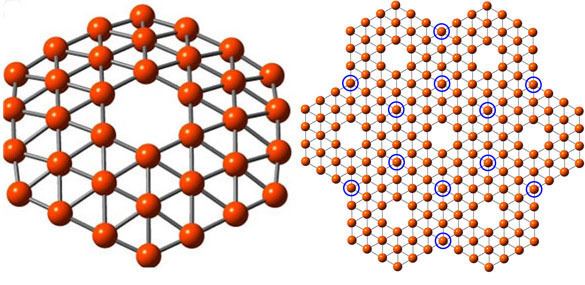
In 2014 a research team at Brown University, led by Lai-Sheng Wang, showed that the structure of B
36 was not only possible but highly stable. Photoelectron spectroscopy revealed a relatively simple spectrum, suggesting a symmetric cluster. Neutral B
36 is the smallest boron cluster to have sixfold symmetry and a perfect hexagonal vacancy, and it can be viewed as a potential basis for extended two-dimensional boron sheets.
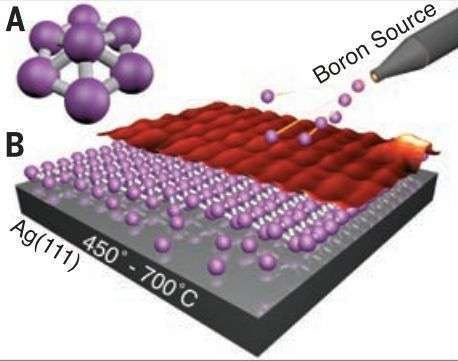
In 2015 a research team synthesized borophene on silver surfaces under ultrahigh-vacuum conditions. Atomic-scale characterization, supported by theoretical calculations, revealed structures reminiscent of fused boron clusters with multiple scales of anisotropic, out-of-plane buckling. Unlike bulk boron allotropes, borophene shows metallic characteristics that are consistent with predictions of a highly anisotropic, 2D metal.
Borospherene
In July 2014 researchers announced the creation of a 40-atom buckyball-like spherical cage made of boron that the team dubbed borospherene (derived from the original "buckminster fullerene".) Where buckyball molecules feature 20 hexagons and 12 pentagons of carbon atoms producing a smooth spherical surface, borospherene consists of 48 triangles, four seven-sided rings and two six-sided rings. The resulting shape is also spherical, but with several atoms sticking out from the sides.
