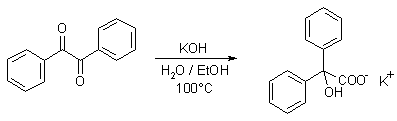Formula C14H12O3 Boiling point 180 °C Appearance white solid | Density 1.08 g/cm³ | |
 | ||
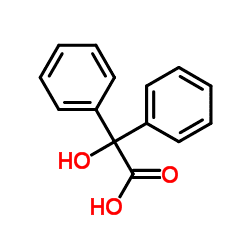
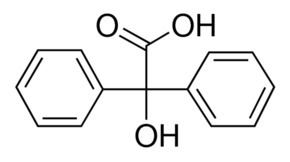
Benzilic acid is an organic compound with formula C
14H
12O
3 or (C
6H
5)2(HO)C(COOH). It is a white crystalline aromatic acid, soluble in many primary alcohols.
Contents
Walsangikar s d 3 benzilic acid rearrangement
Preparation
Benzilic acid can be prepared by heating mixture of benzil, ethanol and potassium hydroxide.
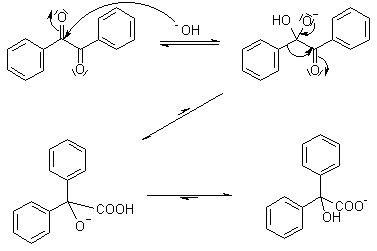
Another preparation, performed by Liebig in 1838, is the dimerization of benzaldehyde, to benzil, which is transformed to the product by the benzilic acid rearrangement reaction.
Uses

Benzilic acid is used in the manufacture of glycollate pharmaceuticals including Clidinium, Dilantin, and Flutropium, which are antagonists of the muscarinic acetylcholine receptors.
It is used in manufacture of the incapacitating agent 3-quinuclidinyl benzilate (BZ) and is regulated by the Chemical Weapons Convention. It is also monitored by law enforcement agencies of many countries, because of its use in the manufacture in hallucinogenic drugs.