 | ||
Avian infectious bronchitis (IB) is an acute and highly contagious respiratory disease of chickens. The disease is caused by avian infectious bronchitis virus (IBV), a coronavirus, and characterized by respiratory signs including gasping, coughing, sneezing, tracheal rales, and nasal discharge. In young chickens, severe respiratory distress may occur. In layers, respiratory distress, nephritis, decrease in egg production, and loss of internal (watery egg white) and external (fragile, soft, irregular or rough shells, shell-less) egg quality are reported.
Contents
Etiology
IBV was the first coronavirus described and varies greatly genetically and phenotypically, with hundreads of serotypes and strains described. Coronaviruses contain the largest known viral RNA genome in number of nucleotides, of approximately 30,000 bases. The RNA forms a single strand and single segment. IBV diversity is based on transcriptional error, which may become very relevant if occurring in genomic sequences coding for proteins, involved in adsorption to target cell or inducing immune responses. Transcriptional error variants may emerge with evolutional advantage in susceptible chickens. Large genomic changes will occur with entire gene interchanges, by reassortment, as for its replication, seven subgenomic mRNAs are produced and will enable reassortment in coinfections.
Clinical signs
Coughing and rattling are common, most severe in young, such as broilers, and rapidly spreading in chickens confined or at proximity. Morbidity is 100% in non-vaccinated flocks. Mortality varies according to the virus strain (up to 60% in non-vaccinated flocks). Respiratory signs will subdue within two weeks. However, for some strains, a kidney infection may follow, causing mortality by toxemia. Younger chickens may die of tracheal occlusion by mucus (lower end) or by kidney failure. The infection may prolong in the cecal tonsils.
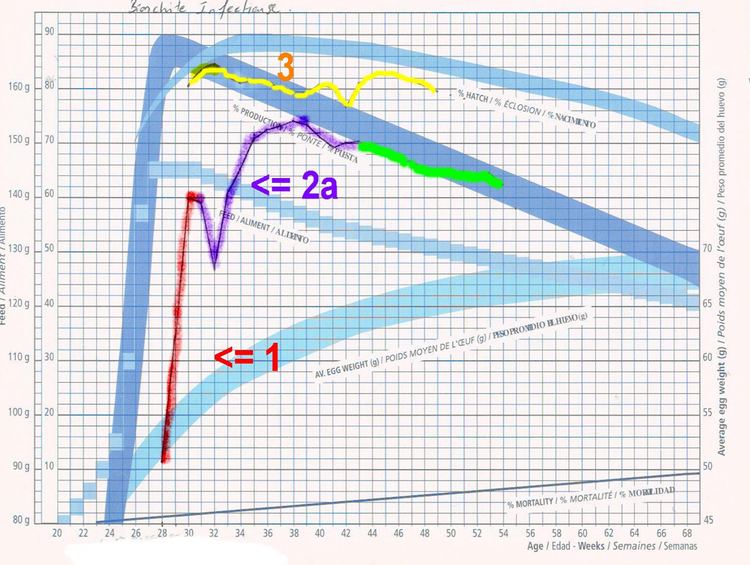
In laying hens, there can be transient respiratory signs, but mortality may be negligible. However, egg production drops sharply. A great percentage of produced eggs are misshapen and discolored. Many laid eggs have a thin or soft shell and poor albumen (watery), and are not marketable or proper for incubation. Normally-colored eggs, indicative of normal shells for instance in brown chickens, have a normal hatchability.
Egg yield curve may never return to normal. Milder strains may allow normal production after around eight weeks.
Read more about clinical AIB here
Diagnosis
Chicken respiratory diseases are difficult to differentiate and may not be diagnosed based on respiratory signs and lesions. Other diseases such as mycoplasmosis by Mycoplasma gallisepticum (chronic respiratory disease), Newcastle disease by mesogenic strains of Newcastle diseases virus (APMV-1), avian metapneumovirus, infectious laryngotracheitis, avian infectious coryza in some stages may clinically resemble IB. Similar kidney lesions may be caused by different etiologies, including other viruses, such as infectious bursal disease virus (the cause of Gumboro disease) and toxins (for instance ochratoxins of Aspergillus ochraceus), and dehydration.
In laying hens, abnormal and reduced egg production are also observed in Egg Drop Syndrome 76 (EDS), cause by an Atadenovirus and avian metapneumovirus infections. At present, IB is more common and far more spread than EDS. The large genetic and phenotypic diversity of IBV have been resulting in common vaccination failures. In addition, new strains of IBV, not present in commercial vaccines, can cause the disease in IB vaccinated flocks. Attenuated vaccines will revert to virulence by consecutive passage in chickens in densely populated areas, and may reassort with field strains, generating potentially important variants.
Definitive diagnosis relies on viral isolation and characterization. For virus characterization, recent methodology using genomic amplification (PCR) and sequencing of products, will enable very precise description of strains, according to the oligonucleotide primers designed and target gene. Methods for IBV antigens detection may employ labelled antibodies, such as direct immunofluorescence or immunoperoxidase. Antibodies to IBV may be detected by indirect immunofluorescent antibody test, ELISA and Haemagglutination inhibition (haemagglutinating IBV produced after enzymatic treatment by phospholipase C).
Treatment and prevention
No specific treatment is available, but antibiotics can be used to prevent secondary infections.
Vaccines are available (ATCvet codes: QI01AA03 (WHO) for the inactivated vaccine, QI01AD07 (WHO) for the live vaccine; plus various combinations).
Biosecurity protocols including adequate isolation, disinfection are important in controlling the spread of the disease.
