Formula C2H7NO2 Melting point 114 °C IUPAC ID Ammonium ethanoate | Molar mass 77.0825 g/mol Density 1.17 g/cm³ Soluble in Water, Methanol | |
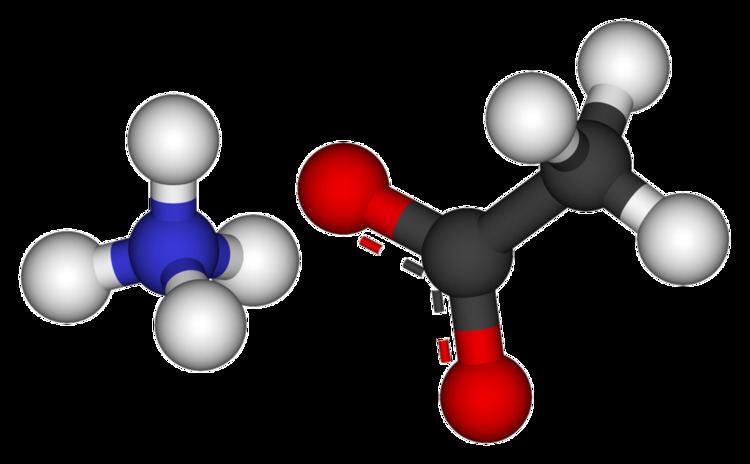 | ||
Appearance White solid crystals, deliquescent | ||
Ammonium acetate
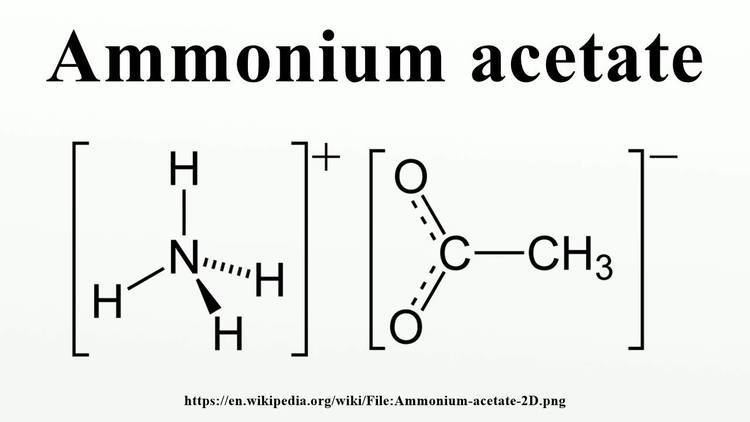
Ammonium acetate is a chemical compound with the formula NH4CH3CO2. It is a white, hygroscopic solid and can be derived from the reaction of ammonia and acetic acid. It is available commercially.
Contents

Ammonium acetate
Uses
It is the main precursor to acetamide:
NH4CH3CO2 → CH3C(O)NH2 + H2OIt is also used as a diuretic.
Buffer
As the salt of a weak acid and a weak base, ammonium acetate is often used with acetic acid to create a buffer solution. Ammonium acetate is volatile at low pressures. Because of this, it has been used to replace cell buffers with non-volatile salts in preparing samples for mass spectrometry. It is also popular as a buffer for mobile phases for HPLC with ELSD detection for this reason. Other volatile salts that have been used for this include ammonium formate.
Other

Food additive

Ammonium acetate is also used as a food additive as an acidity regulator; INS number 264. It is approved for usage in Australia and New Zealand.
Production
Ammonium acetate is produced by the neutralization of acetic acid with ammonium carbonate or by saturating glacial acetic acid with ammonia. Obtaining crystalline ammonium acetate is difficult on account of its aqueous solution giving off ammonia when evaporated.

