Formula C10H14INO2 Melting point 246 °C | Molar mass 307.13 g/mol CAS ID 69587-11-7 | |
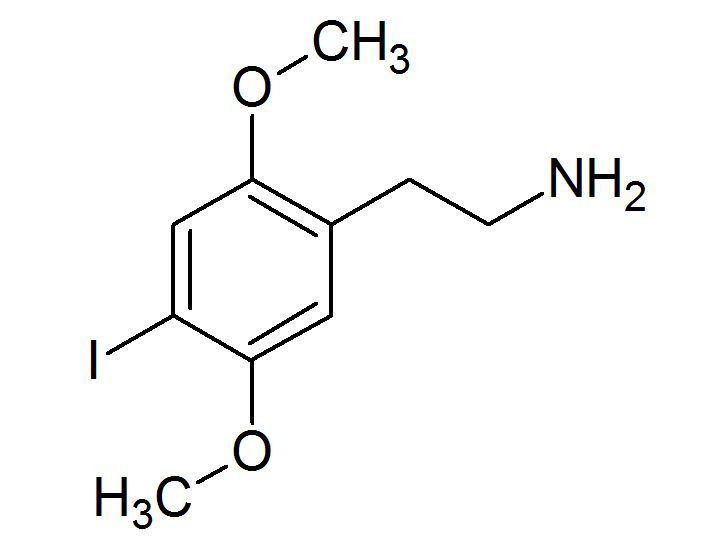 | ||
IUPAC ID 2,5-Dimethoxy-4-iodophenethylamine | ||
Drugtest 2c i with english subtitles
2C-I is a psychedelic phenethylamine of the 2C family. It was first synthesized by Alexander Shulgin and described in his 1991 book PiHKAL: A Chemical Love Story. The drug is used recreationally for its psychedelic and entactogenic effects and is sometimes confused for the analog 25I-NBOMe, nicknamed "Smiles," in the media.
Contents
- Drugtest 2c i with english subtitles
- 2c i what we know
- Recreational use
- European Union
- Canada
- Australia
- Sweden
- United Kingdom
- United States
- References
2c i what we know
Recreational use
In the early 2000s, 2C-I was sold in Dutch smart shops after the drug 2C-B was banned.
According to the US Drug Enforcement Administration, 2C-I is taken orally or snorted in a powder form.
European Union
In December 2003, the European Council issued a binding order compelling all EU member states to ban 2C-I within three months.
Canada
As of October 31st, 2016, 2C-I is a controlled substance (Schedule III) in Canada.
Australia
2C-I is a schedule 9 prohibited substance in Australia under the Poisons Standard (October 2015). A schedule 9 drug is outlined in the Poisons Act 1964 as "Substances which may be abused or misused, the manufacture, possession, sale or use of which should be prohibited by law except when required for medical or scientific research, or for analytical, teaching or training purposes with approval of the CEO."
Sweden
Sveriges riksdag added 2C-I to schedule I ("substances, plant materials and fungi which normally do not have medical use") as narcotics in Sweden as of Mar 16, 2004, published by Medical Products Agency in their regulation LVFS 2004:3 listed as 4-jodo-2,5-dimetoxifenetylamin (2C-I).
United Kingdom
In the United Kingdom, 2C-I is controlled as a Class A substance.
United States
As of July 9, 2012, in the United States 2C-I is a Schedule I substance under the Synthetic Drug Abuse Prevention Act of 2012, making possession, distribution and manufacture illegal. A previous bill, introduced in March 2011, that would have done the same passed the House of Representatives, but was not passed by the Senate.
