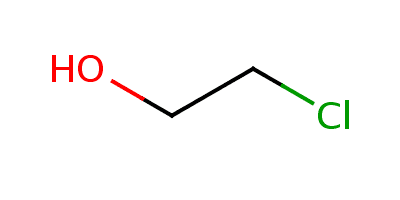
3DMet B01042 Formula C2H5ClO Density 1.2 g/cm³ Melting point -67 °C | Appearance Colourless liquid Boiling point 128.7 °C Molar mass 80.52 g/mol | |
 | ||
Related compounds | ||
Synthesis of 2 chloroethanol
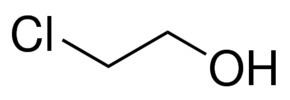
2-Chloroethanol is an chemical compound with the formula HOCH2CH2Cl and the simplest chlorohydrin. This colorless liquid has a pleasant ether-like odor. It is miscible with water. The molecule is bifunctional, consisting of both an alkyl chloride and an alcohol functional groups.
Contents
Synthesis and applications
2-Chloroethanol is produced by treating ethylene with hypochlorous acid:
2-Chloroethanol was once produced on a large scale as a precursor to ethylene oxide:
This application has been supplanted by the greener direct oxidation of ethylene. Otherwise chloroethanol is used in a number of specialized applications. Several dyes are prepared by the alkylation of aniline derivatives with chloroethanol. It is a building block in the production of pharmaceuticals, biocides and plasticizers. It is also used for manufacture of thiodiglycol. It is a solvent for cellulose acetate and ethyl cellulose, textile printing dyes, in dewaxing, refining of rosin, extraction of pine lignin, and the cleaning of machines.
An example is the reaction with theophylline to form Etofylline. Another example is the reaction with chlorphentermine to give Etolorex.
Environmental aspects
Chloroethanol is a metabolite in the degradation of 1,2-dichloroethane. The alcohol is then further oxidized via chloroacetaldehyde to chloroacetate. This metabolic pathway is topical since billions of kilograms of 1,2-dichloroethane are processed annually as a precursor to vinyl chloride.
Safety

2-Chloroethanol is toxic with an LD50 of 89 mg/kg in rats. Like most organochlorine compounds, chloroethanol combusts to yield hydrogen chloride and phosgene.
In regards to dermal exposure to 2-chloroethanol, the Occupational Safety and Health Administration has set a permissible exposure limit of 5 ppm (16 mg/m3) over an eight-hour time-weighted average, while the National Institute for Occupational Safety and Health has a more protective recommended exposure limit of a 1 ppm (3 mg/m3) exposure ceiling.
It is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act (42 U.S.C. 11002), and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.