Boiling point 83.47 °C | Formula C2H4Cl2 Density 1.25 g/cm³ | |
 | ||
Related compounds Thermodynamicdata Phase behavioursolid–liquid–gas | ||
04 01 energy versus dihedral angle 1 2 dichloroethane
The chemical compound 1,2-dichloroethane commonly known as ethylene dichloride (EDC), is a chlorinated hydrocarbon. It is a colourless liquid with a chloroform-like odour. The most common use of 1,2-dichloroethane is in the production of vinyl chloride, which is used to make polyvinyl chloride (PVC) pipes, furniture and automobile upholstery, wall coverings, housewares, and automobile parts.1,2-Dichloroethane is also used generally as an intermediate for other organic chemical compounds and as a solvent. It forms azeotropes with many other solvents, including water (b.p. 70.5 °C) and other chlorocarbons.
Contents
- 04 01 energy versus dihedral angle 1 2 dichloroethane
- Treatment of 1 2 dichloroethane phd vaibhav v gaikwad
- History
- Production
- Vinyl chloride monomer VCM production
- Other uses
- Safety
- References
Treatment of 1 2 dichloroethane phd vaibhav v gaikwad
History
In 1794, physician Jan Rudolph Deiman, merchant Adriaan Paets van Troostwijk, chemist Anthoni Lauwerenburg, and botanist Nicolaas Bondt, under the name of Gezelschap der Hollandsche Scheikundigen (Dutch: Society of Dutch Chemists), were the first to produce 1,2-dichloroethane from olefiant gas (oil-making gas, ethylene) and chlorine gas. Although the Gezelschap in practice did not do much in-depth scientific research, they and their publications were highly regarded. Part of that acknowledgement is that 1,2-dichloroethane was called "Dutch oil" in old chemistry.
Production
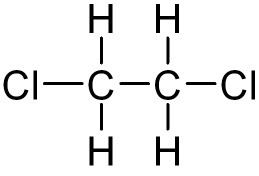
Nearly 20 million tons of 1,2-dichloroethane are produced in the United States, Western Europe, and Japan. Production is primarily achieved through the iron(III) chloride-catalysed reaction of ethene (ethylene) and chlorine.
H2C=CH2 + Cl2 → ClCH2-CH2Cl1,2-dichloroethane is also generated by the copper(II) chloride-catalysed oxychlorination of ethylene:
2 H2C=CH2 + 4 HCl + O2 → 2 ClCH2-CH2Cl + 2 H2OIn principle, it can be prepared by the chlorination of ethane and, less directly, from ethanol.
Vinyl chloride monomer (VCM) production

Approximately 80% of the world's production of 1,2-dichloroethane is used in the production of vinyl chloride monomer (VCM, chloroethene) with hydrogen chloride as a byproduct. VCM is the precursor to polyvinyl chloride.
Cl-CH2-CH2-Cl → H2C=CH-Cl + HClThe hydrogen chloride can be re-used in the production of more 1,2-dichloroethane via the oxychlorination route described above.
Other uses
As a good polar aprotic solvent, 1,2-dichloroethane could be used as degreaser and paint remover but is now banned from use due to its toxicity and possible carcinogenity. As a useful 'building block' reagent, it is used as an intermediate in the production of various organic compounds such as ethylenediamine. In the laboratory it is occasionally used as a source of chlorine, with elimination of ethene and chloride.
Via several steps, 1,2-dichloroethane is a precursor to 1,1,1-trichloroethane, which is used in dry cleaning. Historically, 1,2-dichloroethane was used as an anti-knock additive in leaded fuels.
Safety
1,2-Dichloroethane is toxic (especially by inhalation due to its high vapour pressure), highly flammable, and possibly carcinogenic. Its high solubility and 50-year half-life in anoxic aquifers make it a perennial pollutant and health risk that is very expensive to treat conventionally, requiring a method of bioremediation. While the chemical is not used in consumer products manufactured in the U.S., a case was reported in 2009 of molded plastic consumer products (toys and holiday decorations) from China that released 1,2-dichloroethane into homes at levels high enough to produce cancer risk. Substitutes are recommended and will vary according to application. Dioxolane and toluene are possible substitutes as solvents. Dichloroethane is unstable in the presence of aluminium metal and, when moist, with zinc and iron.
