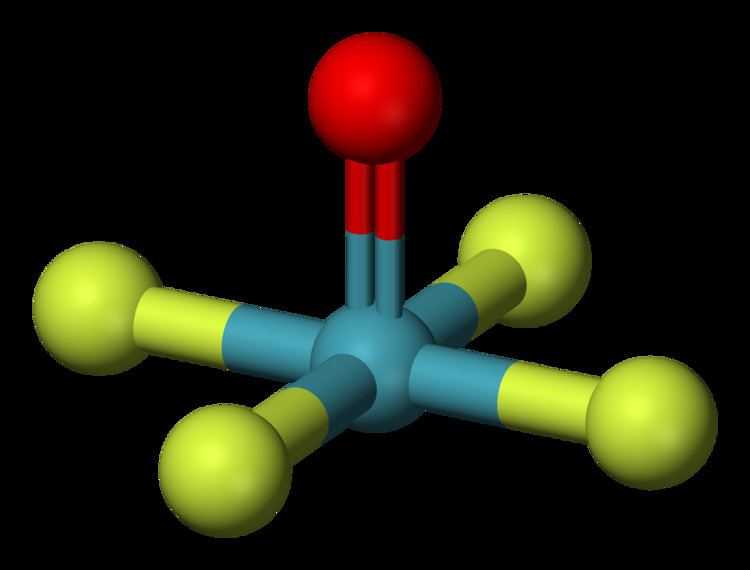
Formula XeOF4 Density 3.17 g/cm³ Melting point -46.2 °C Appearance colorless liquid | Molar mass 223.23 g/mol Boiling point 102 °C Molecular shape square pyramidal | |
 | ||
Similar Xenon trioxide, Xenon hexafluoride, Xenon tetroxide | ||
Lewis dot structure of xeof4 xenon oxytetrafluoride
Xenon oxytetrafluoride (XeOF4) is an inorganic chemical compound. As are most xenon oxides, it is extremely reactive and unstable, and hydrolyses in water to give dangerously hazardous and corrosive products, including hydrogen fluoride:
Contents
In addition, some ozone and fluorine are also formed. The reaction is extremely dangerous, and xenon oxytetrafluoride should therefore be kept away from any trace of water or water vapour under all conditions.
Reactions
XeOF4 reacts with H2O in the following steps:
XeO3 is a dangerous explosive, decomposing explosively to Xe and O2:
References
Xenon oxytetrafluoride Wikipedia(Text) CC BY-SA
