Formula C6O6W Melting point 170 °C Density 2.65 g/cm³ | Molar mass 351.9 g/mol Boiling point 175 °C Appearance Colorless solid | |
 | ||
Tungsten hexacarbonyl (also called tungsten carbonyl) is the chemical compound with the formula W(CO)6. This complex gave rise to the first example of a dihydrogen complex.
Contents
This colorless compound, like its chromium and molybdenum analogs, is noteworthy as a volatile, air-stable derivative of tungsten in its zero oxidation state.
Preparation, properties, and structure
W(CO)6 is prepared by the reduction of WCl6 under a pressure of carbon monoxide. It would be rare to prepare this inexpensive compound in the laboratory because the apparatus is expensive and the compound can be purchased cheaply. The compound is relatively air-stable. It is sparingly soluble in nonpolar organic solvents. Tungsten carbonyl is widely used in electron beam-induced deposition technique - it is easily vaporized and decomposed by the electron beam providing a convenient source of tungsten atoms.
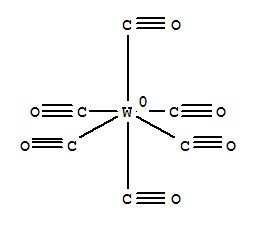
W(CO)6 adopts an octahedral geometry consisting of six rod-like CO ligands radiating from the central W atom with dipole moment 0 D.
Reactivity
All reactions of W(CO)6 commence with displacement of some CO ligands in W(CO)6. W(CO)6 behaves similarly to the Mo(CO)6 but tends to form compounds that are kinetically more robust.
One derivative is the dihydrogen complex W(CO)3[P(C6H11)3]2(H2) reported in 1982 by Kubas.
Three of these CO ligands can be displaced by acetonitrile. W(CO)6 has been used to desulfurize organosulfur compounds and as a precursor to catalysts for alkene metathesis.
Safety and handling
Like all metal carbonyls, W(CO)6 is dangerous source of volatile metal as well as CO.
