UN number 3261 Chemical formula C6H15BF4O | Pubchem 2723982 Molar mass 189.99 g·mol−1 | |
 | ||
Melting point 91 to 92 °C (196 to 198 °F; 364 to 365 K) | ||
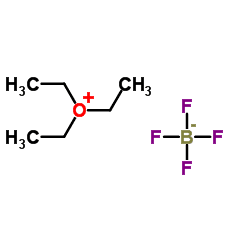
Triethyloxonium tetrafluoroborate is the organic oxonium compound with the formula [(CH3CH2)3O]BF4. It is often called Meerwein's Reagent or Meerwein's Salt after its discoverer Hans Meerwein. Also well known and commercially available is the related trimethyloxonium tetrafluoroborate. The compounds are white solids that dissolve in polar organic solvents. They are strong alkylating agents. Aside from the BF−
4 salt, many related derivatives are available.
Contents
Synthesis and reactivity
Triethyloxonium tetrafluoroborate is prepared from boron trifluoride, diethyl ether, and epichlorohydrin:
4 Et2O·BF3 + 2 Et2O + 3 C2H3(O)CH2Cl → 3 Et3O+BF−4 + B[(OCH(CH2Cl)CH2OEt]3
The trimethyloxonium salt is available from dimethyl ether via an analogous route. These salts do not have long shelf-lives at room temperature. They degrade by hydrolysis:
[(CH3CH2)3O]+BF−4 + H2O → (CH3CH2)2O + CH3CH2OH + HBF4
The propensity of trialkyloxonium salts for alkyl-exchange can be advantageous. For example, trimethyloxonium tetrafluoroborate, which reacts sluggishly due to it low solubility in most compatible solvents may be converted in situ to higher alkyl/more soluble oxoniums, thereby accelerating alkylation reactions.
Structure
The compound features a pyramidal oxonium cation, isoelectronic with triethylamine, and a tetrahedral fluoroborate anion. Reflecting its ionic character, the salt dissolves in polar but inert solvents such as dichloromethane, sulfur dioxide, and nitromethane.
Safety
Triethyloxonium tetrafluoroborate is a strong alkylating agent, although the hazards are diminished because it is non-volatile. It releases strong acid upon contact with water. The properties of the methyl derivative are similar.
