AHFS/Drugs.com Monograph Routes of
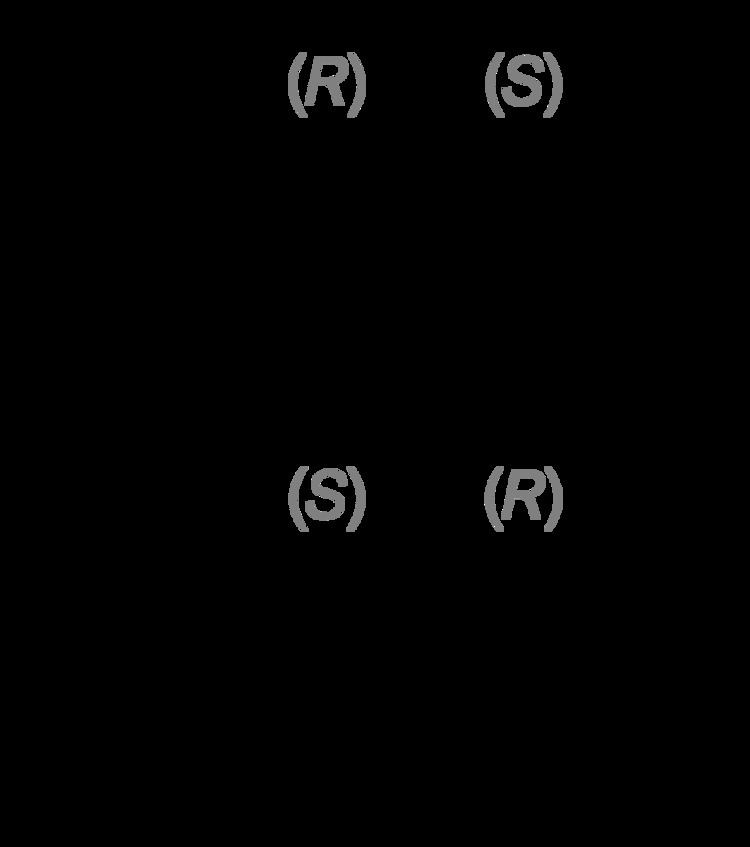
administration Oral CAS ID 155-09-9 Formula C9H11N ATC code N06AF04 (WHO) | MedlinePlus a682088 Molar mass 133.19 g/mol Bioavailability 50% Chirality Racemic mixture | |
 | ||
Trade names originally Parnate, many generics Pregnancy
category AU: B2
US: C (Risk not ruled out) Legal status AU: S4 (Prescription only)
CA: ℞-only
UK: POM (Prescription only)
US: ℞-only
In general: ℞ (Prescription only) Trade name originally Parnate, many generics | ||
Tranylcypromine (INN, USAN, BAN) (originally "Parnate", generic for decades) is a monoamine oxidase inhibitor (MAOI)—it is a nonselective and irreversible inhibitor of the enzyme monoamine oxidase (MAO). It is used as an antidepressant and anxiolytic agent in the clinical treatment of mood and anxiety disorders, respectively.
Contents
Clinical use
Tranylcypromine is used to treat major depressive disorder, especially when there is an anxiety component, typically as a second line treatment.
Contraindications include:
Dietary restrictions
Like other MAOIs, foods high in endogenous monoamine precursors or exogenous monoamine compounds may cause adverse reactions. The most common example, hypertensive crisis, is caused by the ingestion of tyramine, which is found in foods such as aged cheeses, cured meats, tofu and certain red wines. Some, such as yeast extracts, contain enough tyramine to be potentially fatal in a single serving. Spoiled food is also likely to contain dangerous levels of tyramine.
Adverse effects
The most common side effect of tranylcypromine is insomnia; other side effects include orthostatic or postural hypotension, dizziness, drowsiness, fatigue, dry mouth, blurred vision, headache, diarrhea, nausea and vomiting, sleep disturbances, rash and rarely hepatocellular damage, jaundice, hallucinations and blood dyscrasias. Overstimulation including anxiety and agitation, developing rarely into hypomanias; severe hypertensive reactions may occur.
At least one case of the abuse of tranylcypromine has been noted.
Cases of suicidal ideation and suicidal behaviours have been reported during tranylcypromine therapy or early after treatment discontinuation.
Symptoms of tranylcypromine overdose are generally more intense manifestations of its usual effects.
Pharmacology
Tranylcypromine acts as a nonselective and irreversible inhibitor of monoamine oxidase. Regarding the isoforms of monoamine oxidase, it shows slight preference for the MAOB isoenzyme over MAOA. In addition, tranylcypromine functions as a norepinephrine and dopamine releasing agent with approximately 1/10 the potency of amphetamine.
Tranylcypromine has also been shown to inhibit the histone demethylase, BHC110/LSD1. Tranylcypromine inhibits this enzyme with an IC50 < 2 µM, thus acting as a small molecule inhibitor of histone demethylation with an effect to derepress the transcriptional activity of BHC110/LSD1 target genes.
History
Tranylcypromine was originally developed as an analog of amphetamine. Although it was first synthesized in 1948, its MAOI action was not discovered until 1959. Precisely because tranylcypromine was not, like isoniazid and iproniazid, a hydrazine derivative, its clinical interest increased enormously, as it was thought it might have a more acceptable therapeutic index than previous MAOIs.
The drug was introduced by Smith, Kline and French in the United Kingdom in 1960, and approved in the United States in 1961. It was withdrawn from the market in February 1964 due to a number of patient deaths involving hypertensive crises with intracranial bleeding. However, it was reintroduced later that year with more limited indications and specific warnings of the risks.
