The thallium halides include monohalides, where thallium has oxidation state +1, trihalides where thallium generally has oxidation state +3 and some intermediate halides with mixed +1 and +3 oxidation states. These materials find use in specialized optical settings, such as focusing elements in research spectrophotometers. Compared to the more common zinc selenide-based optics, materials such as thallium bromoiodide enable transmission at longer wavelengths. In the infrared, this allows for measurements as low as 350 cm−1 (28 µm), whereas zinc selenide is opaque by 21.5 µm and ZnSe optics are generally only usable to 650 cm−1 (15 µm).
The monohalides all contain thallium with oxidation state +1. Parallels can be drawn between the thallium(I) halides and their corresponding silver salts, for example thallium(I) chloride and bromide are light sensitive and thallium(I) fluoride is more soluble in water than the chloride and bromide.
Thallium(I) fluorideTlF is a white crystalline solid, with a mp of 322 °C. It is readily soluble in water unlike the other
Tl(I) halides. The normal room temperature form has a similar structure to
α-PbO which has a distorted rock salt structure with essentially five coordinate thallium, the sixth fluoride ion is at 370 pm. At 62 °C it transforms to a tetragonal structure. This structure is unchanged up to pressure of 40 GPa.The room temperature structure has been explained in terms of interaction between Tl 6s and the F 2p states producing strongly antibonding Tl-F states. The structure distorts to minimise these unfavourable covalent interactions.
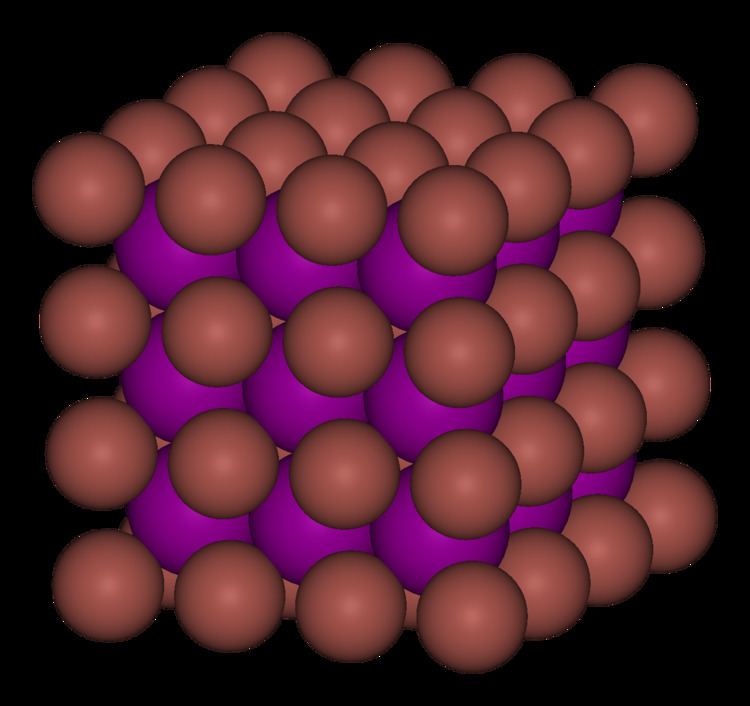
Thallium(I) chlorideTlCl is a light sensitive, white crystalline solid, mp 430 °C. The crystal structure is the same as CsCl.
Thallium(I) bromideTlBr is a light sensitive, pale yellow crystalline solid, mp 460 °C. The crystal structure is the same as CsCl.
Thallium(I) iodideAt room temperature
TlI is a yellow crystalline solid, mp 442 °C. The crystal structure is a distorted rock salt structure known as the
β-TlI structure. At higher temperatures the colour changes to red with a structure the same as CsCl.
Thallium bromoiodide and thallium bromochloride are mixed salts of thallium(I) that are used in spectroscopy as an optical material for transmission, refraction and focusing of infrared radiation. The materials were first grown by R. Koops in the laboratory of Olexander Smakula at the Carl Zeiss Optical Works, Jena in 1941. The red bromoiodide was coded KRS-5 and the colourless bromochloride, KRS-6 and this is how they are commonly known. The KRS prefix is an abbreviation of “Kristalle aus dem Schmelz-fluss”, (crystals from the melt). The compositions of KRS-5 and KRS-6 approximate to TlBr0.4I0.6 and TlBr0.3Cl0.7. KRS-5 is the most commonly used, its properties of being relatively insoluble in water and non-hygroscopic, make it an alternative to KBr, CsI and AgCl.
The thallium trihalides are less stable than their corresponding aluminium, gallium and indium counterparts and chemically quite distinct. The triiodide does not contain thallium with oxidation state +3 but is a thallium(I) compound and contains the linear triiodide (I3−) ion.
Thallium(III) fluorideTlF
3 is a white crystalline solid , mp 550 °C. The crystal structure is the same as YF
3 and
β-BiF3. In this the thallium atom is 9 coordinate,(tricapped trigonal prismatic). It can be synthesised by fluoridation of the oxide, Tl
2O
3, with F
2, BrF
3 or SF
4 at 300 °C.
Thallium(III) chlorideTlCl
3 has a distorted Cr(III) chloride structure like AlCl
3 and InCl
3. Solid TlCl
3 is unstable and disproportionates at 40 °C, losing chlorine to give TlCl. It can be prepared in CH
3CN by treating a solution of TlCl with Cl
2 gas.
Thallium(III) bromideThis unstable compound disproportionates at less than 40 °C to TlBr
2. It can be prepared in CH
3CN by treating a solution of TlBr with Br
2 gas. In water the tetrahydrate complex can be prepared by adding bromine to a stirred suspension of TlBr.
Thallium(I) triiodideTlI3 is a black crystalline solid prepared from
TlI and
I2 in aqueous HI. It does not contain thallium(III), but has the same structure as CsI
3 containing the linear I
3− ion.
As a group these are not well characterised. They contain both Tl(I) and Tl(III), where the thallium(III) atom is present as complex anions e.g. TlCl4−.
TlCl2This is formulated as
TlITlIIICl4.
Tl2Cl3This yellow compound is formulated
TlI3 TlIIICl6.
Tl2Br3This compound is similar to
Tl2Cl3 and is formulated
TlI3TlIIIBr6TlBr2This pale brown solid is formulated
TlITlIIIBr4Tl3I4This compound has been reported as an intermediate in the synthesis of
TlI3 from
TlI and
I2. The structure is not known.
Thallium(I) complexesThallium(I) can form complexes of the type (TlX
3)
2− and (TlX
4)
3− both in solution and when thallium(I) halides are incorporated into alkali metal halides. These doped alkali metal halides have new absorption and emission nbands and are used as phosphors in scintillation radiation detectors.
Thallium(III) fluoride complexesThe salts NaTlF
4 and Na
3TlF
6 do not contain discrete
tetrahedral and
octahedral anions. The structure of NaTlF
4 is the same as fluorite (CaF
2) with Na
I and Tl
III atoms occupying the 8 coordinate Ca
II sites. Na
3TlF
6 has the same structure as
cryolite, Na
3AlF
6. In this the thallium atoms are octahedrally coordinated. Both compounds are usually considered to be mixed salts of Na
+ and Tl
3+.
Thallium(III) chloride complexesSalts of
tetrahedral TlCl
4− and octahedral TlCl
63−are known with various cations.Salts containing TlCl
52− with a square pyramidal structure are known. Interestingly some salts, that nominally contain TlCl
52− actually contain the dimeric anion Tl
2Cl
104−, long chain anions where Tl
III is 6 coordinate and the octahedral units are linked by bridging chlorine atoms, or mixed salts of Tl
IIICl
4 and Tl
IIICl
6.The ion Tl
2Cl
93− where thallium atoms are octahedrally coordinated with three bridging chlorine atoms has been identified in the Caesium salt, Cs
3Tl
2Cl
9.
Thallium(III) bromide complexesSalts of Tl
IIIBr
4− and Tl
IIIBr
63− are known with various cations.The TlBr
52− anion has been characterised in a number of salts and is trigonal bipyramidal. Some other salts that nominally contain TlBr
52− are mixed salts containing TlBr
4− and Br
−.
Thallium(III) iodide complexesSalts of
TlIIII4− are known. The
TlIII anion is stable even though the triiodide is a thallium(I) compound.