Abbreviations TMS Boiling point 26.5 °C Appearance Colourless liquid | Formula C4H12Si Density 648 kg/m³ | |
 | ||
Related silanes Related compounds | ||
21 1 explain the use of tetramethylsilane tms as the reference standard hl ib chemistry
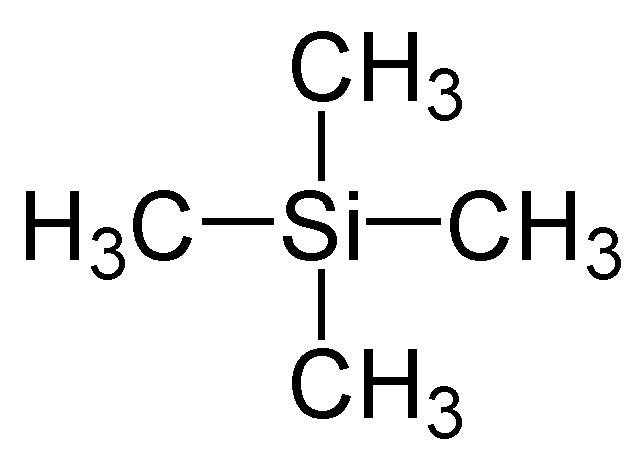
Tetramethylsilane (abbreviated as TMS) is the organosilicon compound with the formula Si(CH3)4. It is the simplest tetraorganosilane. Like all silanes, the TMS framework is tetrahedral. TMS is a building block in organometallic chemistry but also finds use in diverse niche applications.
Contents
- 21 1 explain the use of tetramethylsilane tms as the reference standard hl ib chemistry
- Synthesis and reaction
- Uses in NMR spectroscopy
- References

Synthesis and reaction
TMS is a by-product of the production of methyl chlorosilanes, SiClx(CH3)4−x, via the direct process of reacting methyl chloride with silicon. The more useful products of this reaction are those for x = 1, 2, and 3.
TMS undergoes deprotonation upon treatment with butyllithium to give (H3C)3SiCH2Li. The latter, trimethylsilylmethyl lithium, is a relatively common alkylating agent.

In chemical vapor deposition, TMS is the precursor to silicon dioxide or silicon carbide, depending on the deposition conditions.
Uses in NMR spectroscopy
Tetramethylsilane is the accepted internal standard for calibrating chemical shift for 1H, 13C and 29Si NMR spectroscopy in organic solvents (where TMS is soluble). In water, where it is not soluble, sodium salts of DSS, 2,2-dimethyl-2-silapentane-5-sulfonate, are used instead. Because of its high volatility, TMS can easily be evaporated, which is convenient for recovery of samples analyzed by NMR spectroscopy.
Because all twelve hydrogen atoms in a tetramethylsilane molecule are equivalent, its 1H NMR spectrum consists of a singlet. The chemical shift of this singlet is assigned as δ 0, and all other chemical shifts are determined relative to it. The majority of compounds studied by 1H NMR spectroscopy absorb downfield of the TMS signal, thus there is usually no interference between the standard and the sample. Similarly, all four carbon atoms in a tetramethylsilane molecule are equivalent. In a fully decoupled 13C NMR spectrum, the carbon in the tetramethylsilane appears as a singlet, allowing for easy identification. The chemical shift of this singlet is also set to be δ 0 in the 13C spectrum, and all other chemical shifts are determined relative to it.
Commercial NMR solvents often are supplied without TMS. 1H NMR spectra can be calibrated against residual protio-solvent (e.g. the remaining 0.001% or so of undeuterated chloroform in commercial CDCl3). As deuterium is not observed in 1H NMR, the residual protio-solvent signals can be observed clearly. For 13C NMR work, spectra are usually calibrated against the deuterated solvent peak. For example, deuterochloroform shows a triplet of equal height at δ 77.0. The triplet is explained by applying the 2nI + 1 rule; for the case of deuterium, I = 1. Tables and charts of chemical shifts for various types of NMR spectroscopy are often provided by vendors of NMR solvents. Work has also been done to prepare comprehensive tables of chemical shifts of solvents and impurities.
