 | ||
The terpinenes are a group of isomeric hydrocarbons that are classified as terpenes. They each have the same molecular formula and carbon framework, but they differ in the position of carbon-carbon double bonds. α-Terpinene has been isolated from cardamom and marjoram oils, and from other natural sources. β-Terpinene has no known natural source, but has been prepared synthetically from sabinene. γ-Terpinene and δ-terpinene (also known as terpinolene) are natural and have been isolated from a variety of plant sources.
Contents
- What does terpinene mean
- Uses
- Biosynthesis of terpinene
- List of the plants that contain one of the chemicals
- References
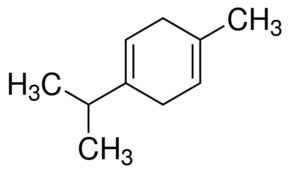
What does terpinene mean
Uses
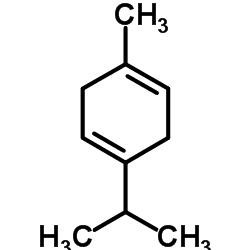
α-Terpinene is a perfume and flavoring chemical used in the cosmetics and food industries. Its use in both the pharmaceutical and the electronics semi-conductor manufacturing industries has also proven to be valuable.
Biosynthesis of α-terpinene
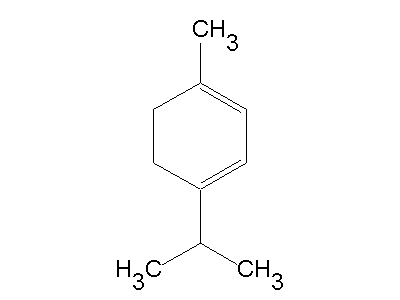
The biosynthesis of α-terpinene and other terpenoids occurs via the mevalonate pathway because its starting reactant, dimethylallyl pyrophosphate (DMAPP), is derived from mevalonic acid.
Geranyl pyrophosphate (GPP) is produced from the reaction of a resonance-stable allylic cation, formed from the loss of the pyrophosphate group from DMAPP, and isopentenyl pyrophosphate (IPP), and the subsequent loss of a proton. GPP then loses the pyrophosphate group to form the resonance-stable geranyl cation. The reintroduction of the pyrophosphate group to the cation produces GPP isomer, known as linalyl pyrophosphate (LPP). LPP then forms a resonance-stable cation by losing its pyrophosphate group. Cyclization is then completed thanks to this more favorable stereochemistry of the LPP cation, now yielding a terpinyl cation. Finally, a 1,2-hydride shift via a Wagner-Meerwein rearrangement produces the terpinen-4-yl cation. It is the loss of a hydrogen from this cation that generates α-terpinene.
