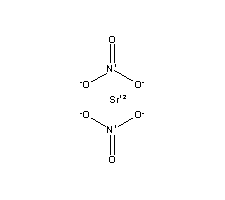
Formula Sr(NO3)2 Melting point 570 °C Boiling point 645 °C | Molar mass 211.63 g/mol Density 2.99 g/cm³ Appearance white granular solid | |
 | ||
Testing strontium nitrate red fountain composition
Strontium nitrate is an inorganic compound with the formula Sr(NO3)2. This colourless solid is used as a colorant (red) in pyrotechnics and is also used as an oxidizer in pyrotechnics.
Contents
- Testing strontium nitrate red fountain composition
- Strontium nitrate reaction only
- Preparation
- Uses
- Biochemistry
- Popular Culture
- References
Strontium nitrate reaction only
Preparation
Strontium nitrate is typically generated by the reaction of nitric acid on strontium carbonate.
2 HNO3 + SrCO3 → Sr(NO3)2 + H2O + CO2.
Uses
Like many other strontium salts, strontium nitrate is used to produce a rich red flame in fireworks and road flares. The oxidizing properties of this salt are advantageous in such applications.
Strontium nitrate can aid in eliminating and lessening skin irritations. When mixed with glycolic acid, strontium nitrate reduces the sensation of skin irritation significantly better than using glycolic acid alone.
Biochemistry
As a divalent ion with an ionic radius similar to that of Ca2+ (1.13 vs. 0.99 A, respectively), Sr2+ ions resembles calcium's ability to traverse calcium-selective ion channels and trigger neurotransmitter release from nerve endings. It is thus used in electrophysiology experiments.
Popular Culture
In his short story "A Germ Destroyer," Rudyard Kipling refers to strontium nitrate as the main ingredient of the titular fumigant
