CAS Number 16037-91-5 CAS ID 16037-91-5 | Molar mass 910.9 g/mol | |
 | ||
Trade names Pentostam, Stiboson, others AHFS/Drugs.com International Drug Names Routes ofadministration intravenous, intramusclar ATC code P01CB02 (WHO) QP51AB02 (WHO) Legal status UK: POM (Prescription only) | ||
Sodium stibogluconate, sold under the brand name Pentostam among others, is a medication used to treat leishmaniasis. This includes leishmaniasis of the cutaneous, visceral, and mucosal types. Some combination of miltefosine, paramycin and liposomal amphotericin B, however, may be recommended due to issues with resistance. It is given by injection.
Contents
Side effects are common and include loss of appetite, nausea, muscle pains, headache, and feeling tired. Serious side effect may include an irregular heartbeat or pancreatitis. Sodium stibogluconate is less safe than some other options during pregnancy. It is not believed to result in any problems if used during breastfeeding. Sodium stibogluconate is in the pentavalent antimonials class of medication.

Sodium stibogluconate has been studied as early as 1937 and has been in medical use since the 1940s. It is on the World Health Organization's List of Essential Medicines, the most effective and safe medicines needed in a health system. The wholesale price in the developing world is 9.66 USD per vial. This gives the a for a course of treatment at between 12 and 56 USD. In the United States it is available from the Center for Disease Control.
Side effects
Sodium stibogluconate is exceedingly toxic to veins. One of the practical problems is that after a few doses it can become exceedingly difficult to find a vein in which to inject the drug. The insertion of a PICC does not prevent the problem and can instead exacerbate it: the entire vein along the course of the PICC line can become inflamed and thrombose. Large doses of sodium stibogluconate are often administered as dilute solutions.
Pancreatitis is a common deleterious effect of the drug, and the serum amylase or lipase should be monitored twice weekly; there is no need to stop treatment if the amylase remains less than four times the upper limit of normal; if the amylase rises above the cut-off, then treatment should be interrupted until the amylase falls to less than twice the upper limit of normal, whereupon treatment can be resumed. Cardiac conduction disturbances are less common, but ECG monitoring while the medicine is injected is advisable and changes quickly reverse after the drug is stopped or the infusion rate is decreased.
The drug can be given intramuscularly but is exceedingly painful when given by this route. It can also be given intralesionally when treating cutaneous leishmaniasis (i.e., injected directly into the area of infected skin) and again, this is exceedingly painful and does not give results superior to intravenous administration.
Sodium stibogluconate can also cause a reduced appetite, metallic taste in mouth, nausea, vomiting, diarrhoea, headache, tiredness, joint pains, muscle aches, dizziness, and anaphylaxis.
Dosing
Sodium stibogluconate is available in the United Kingdom as Pentostam, where it is manufactured by GlaxoSmithKline. It is available in the United States on a named-patient basis from the Centers for Disease Control and Prevention (CDC).
The dose of sodium stibogluconate is by slow intravenous infusion (at least five minutes with cardiac monitoring). The injection are stopped if there is coughing or central chest pain. The chemotherapeutic index was established by Leonard Goodwin during the Second World War when a treatment was urgently required for Allied troops during the invasion of Sicily.
The duration of treatment is usually 10 to 21 days and depends on the species of Leishmania and the type of infection (cutaneous or visceral).
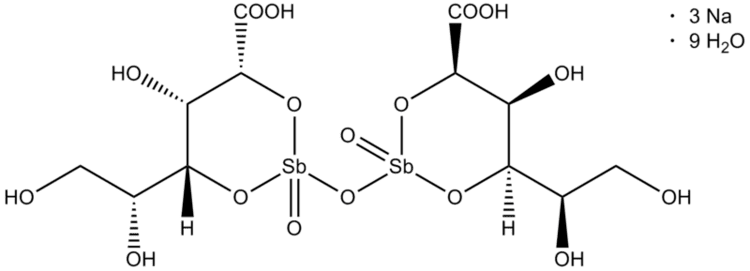
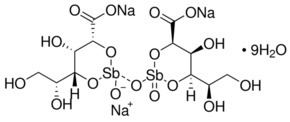
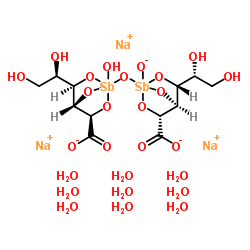
Chemical structure
The chemical structure of sodium stibogluconate is somewhat ambiguous, and the structure shown above is idealized. Its solutions may contain multiple antimony compounds, although this heterogeneity may be unimportant. It has been speculated that the active species contains only a single antimony centre.
Pharmacokinetics
Although antimony itself is a cause of heavy metal poisoning, stibogluconate does not appear to accumulate in the body and is excreted by the kidneys.
