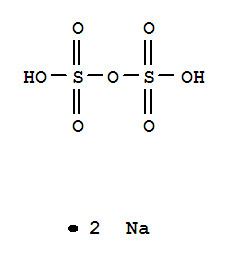
Formula Na2S2O7 Density 2.66 g/cm³ Melting point 400.9 °C | Molar mass 222.12 g/mol Boiling point 460 °C | |
 | ||
Appearance Translucent white crystals | ||
Sodium pyrosulfate, or Disodium disulfate, is an inorganic compound with the chemical formula is Na2S2O7.
Contents
Preparation
Sodium pyrosulfate is obtained by the dehydration of sodium bisulfate:
2 NaHSO4 → Na2S2O7 + H2OTemperatures above 460 °C further decompose the compound, producing sodium sulfate and sulfur trioxide:
Na2S2O7 → Na2SO4 + SO3Applications
Sodium pyrosulfate is used in analytical chemistry; samples are fused with sodium pyrosulfate to ensure complete dissolution prior to a quantitative analysis.
References
Sodium pyrosulfate Wikipedia(Text) CC BY-SA
