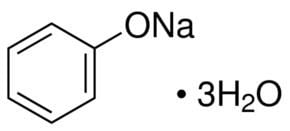
Molar mass 116.09 g/mol | Appearance White solid | |
 | ||
Sodium phenoxide is an organic compound with the formula NaOC6H5. This white solid is the conjugate base of phenol. It is used as a precursor to many other organic compounds, such as aryl ethers.
Contents
Synthesis and structure
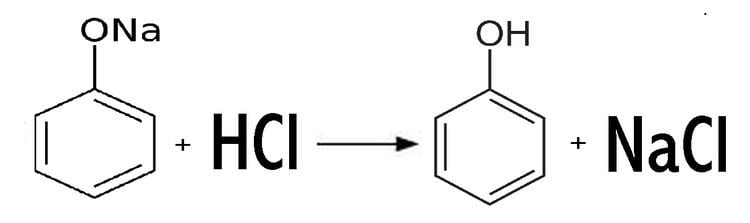
Most commonly, solutions of sodium phenoxide are produced by treating phenol with sodium hydroxide. Anhydrous derivatives can be prepared from phenol and sodium:
Na + HOC6H5 → NaOC6H5 + 1/2 H2
Like other sodium alkoxides, crystalline sodium phenolate adopts a complex structures involving multiple Na-O bonds. Solvent-free material is polymeric, each Na center being bound to three oxygen ligands as well as the phenyl ring. Adducts of sodium phenoxide are molecular, such as the cubane [NaOPh]4(HMPA)4.
Sodium phenoxide is produced by the "alkaline fusion" of benzenesulfonic acid, whereby the sulfonate groups is displaced by hydroxide:
C6H5SO3Na + 2 NaOH → C6H5ONa + Na2SO3This route once was the principal industrial route to phenol.
Reactions
Sodium phenoxide is a moderately strong base. At low pH's gives phenol:
PhOH ⇌ PhO− + H+ (K = 10−10)Sodium phenoxide can be used to prepare phenyl ethers and metal phenolates:
NaOC6H5 + RBr → ROC6H5 + NaBr