Appearance white powder Molar mass 209.94 g/mol Density 2.9 g/cm³ | Formula Na3AlF6 Melting point 1,000 °C | |
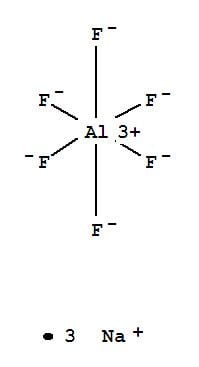 | ||
Sodium aluminium hexafluoride is the inorganic compound with the formula Na3AlF6. This white solid occurs naturally as the mineral cryolite and is used extensively in the industrial production of aluminium metal. The compound consists of the sodium (Na+) salt of hexafluoroaluminate (AlF63−).
Contents
Production
Most cryolite is manufactured from aluminium oxides, hydrofluoric acid, and sodium hydroxide or the equivalent reagent hexafluorosilicic acid:
6 NaOH + Al2O3 + 12 HF → 2 Na3AlF6 + 9 H2OIt was mined as cryolite at Ivigtût on the west coast of Greenland, until the deposit was depleted in 1987.
Use
The main application of synthetic cryolite is as a solvent (or flux) for electrolysis aluminium oxides such as bauxite. The conversion of aluminium oxides into metallic aluminium requires that the metal ions be dissolved so that they can accept the electrons provided in the electrolysis cell. A mixture of cryolite and some aluminium trifluoride is that solvent. Unlike typical solutions, this one requires temperatures approaching 1000 °C to melt. Sodium aluminium hexafluoride is also used as a pesticide. Other uses include a whitener for enamels and an opacifier for glass.
Safety
Cryolite is poorly soluble in water, which mitigates problems. The LD50 = 600 mg/kg for the comparable compound aluminium trifluoride.
