Formula NaAlH4 Appearance White crystalline solid | Density 905 kg/m³ | |
 | ||
Sodium aluminium hydride or sodium alanate is an inorganic compound with the chemical formula NaAlH4. It is a white pyrophoric solid that dissolves in tetrahydrofuran (THF), but not in diethyl ether or hydrocarbons. It has been evaluated as an agent for the reversible storage of hydrogen and it is used as a reagent for the chemical synthesis of organic compounds. Similar to lithium aluminium hydride, it is a salt consisting of separated sodium cations and tetrahedral AlH−
4 anions.
Contents
Structure, preparation, and reactions
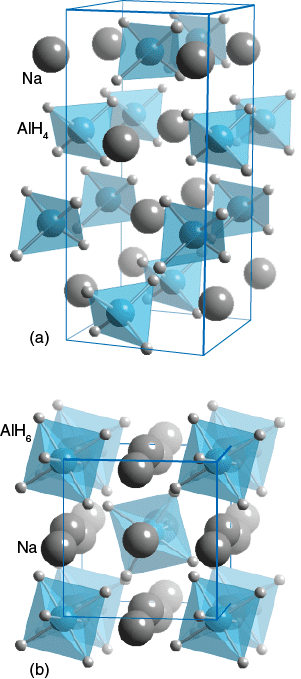
Sodium tetrahydroaluminate adopts the structure of (is isostructural with) calcium tungstate. As such, the tetrahedral AlH−
4 centers are linked with eight-coordinat Na+ cations. The compound is prepared from the elements under high pressures of H2 at 200 °C using triethylaluminium catalyst:
As a suspension in diethyl ether, it reacts with lithium chloride to give the popular reagent lithium aluminium hydride:
LiCl + NaAlH4 → LiAlH4 + NaClThe compound reacts rapidly, even violently, with protic reagents, such as water, as described in this idealized equation:
4 H2O + NaAlH4 → "NaAl(OH)4" + 4 H2Hydrogen storage
Sodium alanate has been explored for hydrogen storage in hydrogen tanks. The relevant reactions are:
3 NaAlH4 → Na3AlH6+ Al + H2Na3AlH6 → 3 NaH + Al + 3/2 H2Sodium tetrahydroaluminate can release up to 7.4 wt % of hydrogen when heated at 200 °C (392 °F). Absorption can be slow, with several minutes being required to fill a tank. Both release and uptake are catalysed by titanium.
Reagent in organic chemistry
Sodium aluminium hydride is a strong reducing agent, very similar in reactivity to lithium aluminum hydride (LAH) and, to some extent, Diisobutylaluminium hydride (DIBAL) in organic reactions. It is much more powerful reducing agent than sodium borohydride due to the weaker and more polar Al-H bond compared to the B-H bond. Like LAH, it reduces esters to alcohols.
Safety
Sodium aluminium hydride is highly flammable. It does not react in dry air at room temperature but is very sensitive to moisture. It ignites or explodes on contact with water.
