Formula Ag2S Density 7.23 g/cm³ | Molar mass 247.8 g/mol Appearance Grayish-black crystal | |
 | ||
The solubility of silver sulfide and the effect of ph
Silver sulfide (Ag
2S) is the sulfide of silver. It is useful as a photosensitizer in photography.
Contents
Properties
This dense black solid constitutes the tarnish that forms over time on silverware and other silver objects. Silver sulfide is insoluble in all solvents, but is degraded by strong acids. Silver sulfide is an ionic bond, as it is made up of silver (electronegativity of 1.98) and sulfur (electronegativity of 2.58). It is a component of classical qualitative inorganic analysis. When formed on electrical contacts operating in an atmosphere rich in hydrogen sulfide, long filaments known as silver whiskers can form.
Degrading wooden treasure chests aboard sunken galleons can provide the sulfide needed for certain sulfide ion consuming bacteria to produce hydrogen sulfide gas. When combined with silver the hydrogen sulfide gas creates a layer of black silver sulfide patina on the silver, protecting the inner silver from further conversion to silver sulfide.
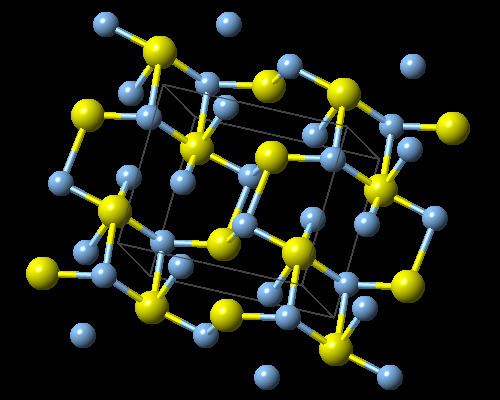
Structure
Three forms are known: monoclinic acanthite (β-form), stable below 179 °C, body centered cubic so-called argentite (α-form), stable above 180 °C, and a high temperature face-centred cubic (γ-form) stable above 586 °C. The higher temperature forms are electrical conductors. It is found in nature as relatively low temperature mineral acanthite. Acanthite is an important ore of silver. In the acanthite, monoclinic, form there are two crystallographically distinct silver atoms with two and three near neighbour sulfur atoms respectively. The name argentite refers to a cubic form, which, due to instability in "normal" temperatures, is found in form of the pseudomorphosis of acanthite after argentite.
