Formula Ag2C2O4 Density 5 g/cm³ Appearance white powder | Molar mass 303.755 g/mol Melting point 140 °C | |
 | ||
Ferrocerium and silver oxalate
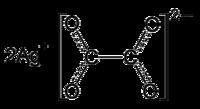
Silver oxalate (Ag
2C
2O
4) is commonly employed in experimental petrology to add carbon dioxide (CO
2) to experiments as it will break down to silver (Ag) and carbon dioxide under geologic conditions. It is also a precursor to the production of silver nanoparticles. It is explosive upon heating around 140 degrees Celsius, shock or friction.
Contents
Production
Silver oxalate is produced by the reaction between silver nitrate and oxalic acid.
References
Silver oxalate Wikipedia(Text) CC BY-SA
