Formula H4P2O7 Melting point 71.5 °C | Molar mass 177.98 g/mol Pubchem 1023 | |
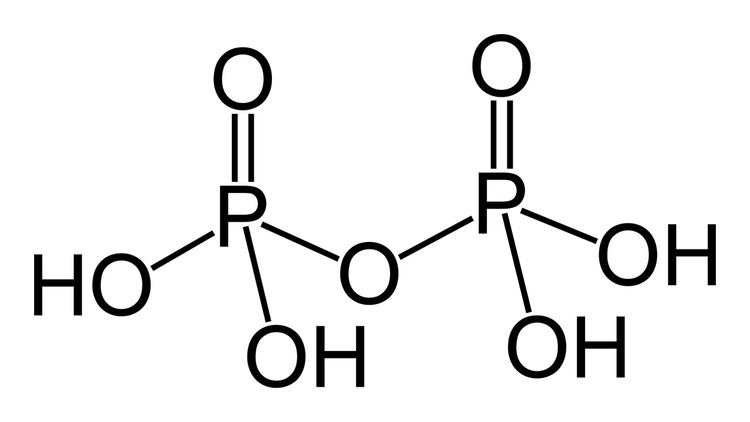 | ||
Pyrophosphoric acid meaning
Pyrophosphoric acid, also known under the name diphosphoric acid, is colorless, odorless, hygroscopic and is soluble in water, diethyl ether, and ethyl alcohol. The anhydrous acid has two different forms, which melt at 54.3 °C and 71.5 °C. It is best prepared by ion exchange from sodium pyrophosphate or by reacting hydrogen sulfide with lead pyrophosphate. When phosphoric acid is dehydrated pyrophosphoric acid is produced as one of the products. When molten pyrophosphoric acid rapidly forms an equilibrium mixture of phosphoric acid, pyrophosphoric acid and polyphosphoric acids. The percentage by weight of pyrophosphoric acid is around 40% and it is difficult to recrystallise from the melt. In aqueous solution pyrophosphoric acid like all polyphosphoric acids hydrolyses and eventually an equilibrium is established between phosphoric acid pyrophosphoric acid and polyphosphoric acids. When highly diluted an aqueous solution of pyrophosphoric acid contains only phosphoric acid.
Contents
Pyrophosphoric acid is a medium strong inorganic acid. Anions, salts, and esters of pyrophosphoric acid are called pyrophosphates.
While pyrophosporic acid is corrosive, it is not known to be otherwise toxic.
What does pyrophosphoric acid mean
History
The name pyrophosphoric acid was given by a "Mr. Clarke of Glasgow" in 1827 who is credited with its discovery following the heating to red heat of a sodium phosphate salt. It was found that phosphoric acid when heated to red heat formed pyrophosphoric acid that was readily converted to phosphoric acid by hot water.
