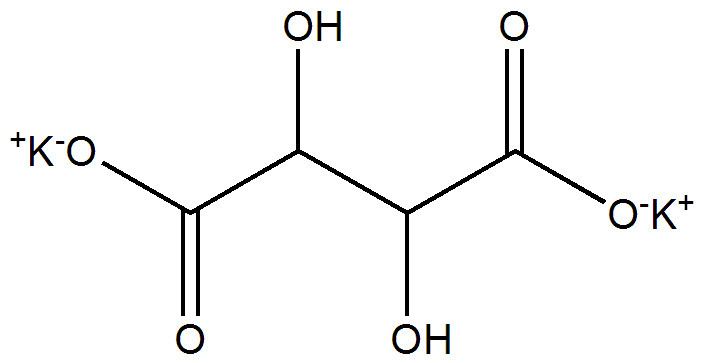
Formula C4H4K2O6 Density 1.984 g/cm³ | Molar mass 226.27 g/mol | |
 | ||
Appearance colorless, slightly opaque crystals | ||
Catalyzed oxidation of sodium potassium tartrate
Potassium tartrate, dipotassium tartrate or argol has formula K2C4H4O6. It is the potassium salt of tartaric acid. It is often confused with potassium bitartrate, also known as cream of tartar. As a food additive, it shares the E number E336 with potassium bitartrate.
Contents
- Catalyzed oxidation of sodium potassium tartrate
- Potassium tartrate solution is added to water callie
- Manufacturing
- Other compounds
- References
Potassium tartrate solution is added to water callie
Manufacturing
Potassium tartrate is produced by the reaction of tartaric acid with potassium sodium tartrate (rochelle salt), and potassium sulfate, followed by filtration, purification, precipitation and drying.
Other compounds
Tartar emetic is produced when potassium tartrate is heated with antimony trioxide. Tartar emetic causes intense nausea, prostration and vomiting by irritating the gastrointestinal mucosa.
References
Potassium tartrate Wikipedia(Text) CC BY-SA
