Formula C20H14N4 Appearance Dark red, shiny leaflets | Molar mass 310.352 g/mol | |
 | ||
Calcium carbonate and porphin reaction
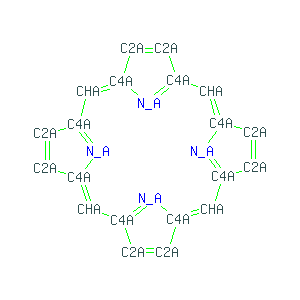
Porphin, sometimes spelled porphine, is the parent chemical compound for types of biochemically significant compounds called porphyrins. The chemical formula of porphin is C20H14N4. Porphin is an organic compound that is aromatic and heterocyclic since its chemical structure, shown at right, essentially consists of four pyrrole rings joined together by four methine (=CH—) groups to form a larger macrocycle ring. The compound itself is a solid.
Contents
- Calcium carbonate and porphin reaction
- How to make porphin from chlorophyll
- Structural characteristics of porphin
- Complex with metal in the center
- References
How to make porphin from chlorophyll
Structural characteristics of porphin
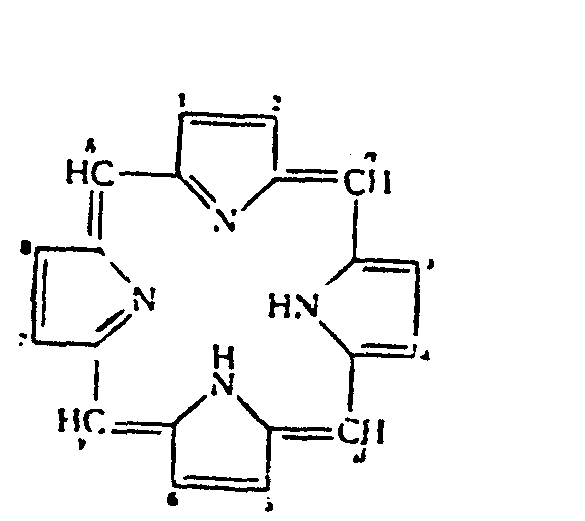
Around the perimeter of the macrocycle ring, there is a cyclic chain of sp2 hybridized carbon atoms, all of which are part of a conjugated double bond system, giving the molecule its aromatic character. Resonance structures for porphin can be drawn as shown below here.
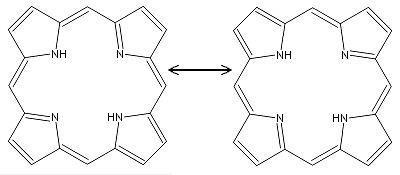
The aromatic character of porphin stems both from its conjugation as well as its flat or planar geometry, meaning that all the atoms lie in a single plane. Bonded to the cyclic chain of carbons are four nitrogen atoms facing the center of the molecule, two of which are bonded to hydrogen atoms and the other two nitrogens forming part of the conjugated double bond system.
Complex with metal in the center

If the two nitrogens give up their hydrogens, the four central nitrogen atoms can act as ligands to bond to a metal ion in the center of the molecule. This is often the case with biological porphyrin compounds, especially with an iron atom in the center. When there is no metal ion (or atom) bound to the nitrogens in the center, then the compounds are called free porphin or free porphyrins. If they are bonded to a metal in the center, then they are bound. A porphyrin with an iron atom of the type found in myoglobin, hemoglobin, or certain cytochromes is called heme. See the Porphyrin article for further details.

Compounds similar to porphin are the parent compounds for similar ring systems with other central metal atoms in biochemistry. These include chlorin, which has a magnesium atom in several types of chlorophyll; bacteriochlorin, found in bacteriochlorophyll; and corrin, which has a cobalt atom in cobalamin or vitamin B12.