Trade names Pomalyst, Imnovid Routes ofadministration Oral (capsules) Molar mass 273.24 g/mol | AHFS/Drugs.com Monograph ATC code L04AX06 (WHO) | |
 | ||
License data EU EMA: ImnovidUS FDA: Pomalidomide Pregnancycategory AU: X (High risk)US: X (Contraindicated) | ||
Pomalidomide in multiple myeloma
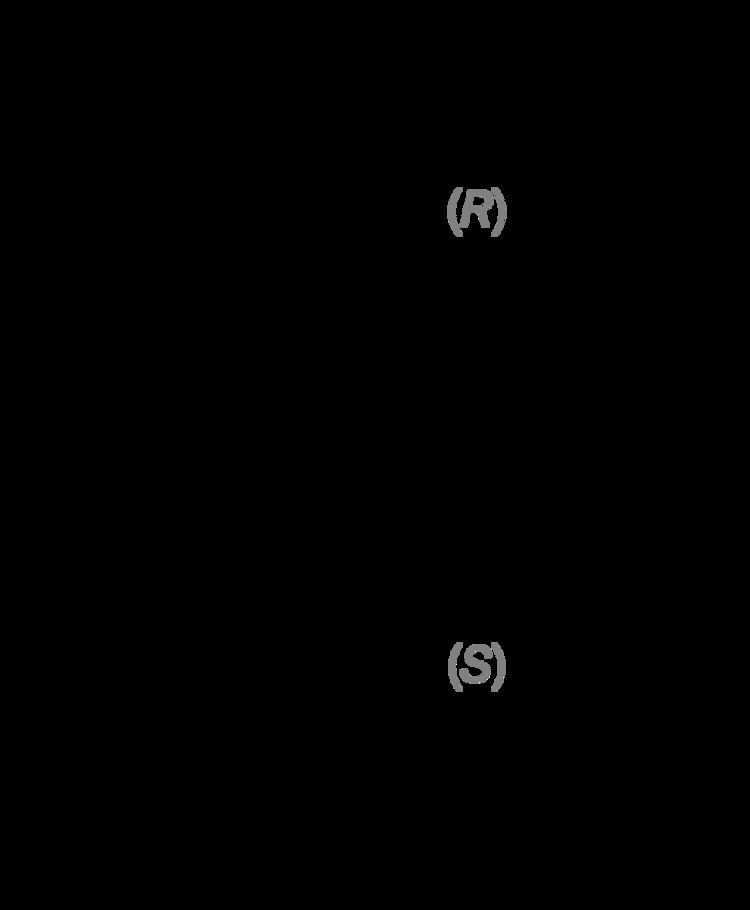
Pomalidomide (INN, codenamed CC-4047, also known as 3-amino-thalidomide, trade name Pomalyst in the U.S. and Imnovid in EU and Russia) is a derivative of thalidomide marketed by Celgene. It is anti-angiogenic and also acts as an immunomodulator.
Contents
- Pomalidomide in multiple myeloma
- Eha 2013 pomalidomide prolongs survival in refractory multiple myeloma dr jesus san miguel
- Origin and development
- Clinical trials
- Mechanism
- Pregnancy and sexual contact warnings
- References
Pomalidomide was approved in February 2013 by the U.S. Food and Drug Administration (FDA) as a treatment for relapsed and refractory multiple myeloma. It has been approved for use in people who have received at least two prior therapies including lenalidomide and bortezomib and have demonstrated disease progression on or within 60 days of completion of the last therapy. It received a similar approval from the European Commission in August 2013.
Eha 2013 pomalidomide prolongs survival in refractory multiple myeloma dr jesus san miguel
Origin and development
The parent compound of pomalidomide, thalidomide, was originally discovered to inhibit angiogenesis in 1994. Based upon this discovery, thalidomide was taken into clinical trials for cancer, leading to its ultimate FDA approval for multiple myeloma. Structure-activity studies revealed that amino substituted thalidomide had improved antitumor activity, which was due to its ability to directly inhibit both the tumor cell and vascular compartments of myeloma cancers. This dual activity of pomalidomide makes it more efficacious than thalidomide in vitro and in vivo.
Clinical trials
Phase I trial results showed tolerable side effects.
Phase II clinical trials for multiple myeloma and myelofibrosis reported 'promising results'.
Phase III results showed significant extension of progression-free survival, and overall survival (median 11.9 months vs. 7.8 months; p = 0.0002) in patients taking pomalidomide and dexamethasone vs. dexamethasone alone.
Mechanism
Pomalidomide directly inhibits angiogenesis and myeloma cell growth. This dual effect is central to its activity in myeloma, rather than other pathways such as TNF alpha inhibition, since potent TNF inhibitors including rolipram and pentoxifylline do not inhibit myeloma cell growth or angiogenesis. Upregulation of interferon gamma, IL-2 and IL-10 as well as downregulation of IL-6 have been reported for pomalidomide. These changes may contribute to pomalidomide's anti-angiogenic and anti-myeloma activities.
Pregnancy and sexual contact warnings
Because pomalidomide can cause harm to unborn babies when administered during pregnancy, women taking pomalidomide must not become pregnant. To avoid embryo-fetal exposure, a "Risk Evaluation and Mitigation Strategy" (REMS) program was developed to ensure pregnancy prevention or distribution of the drug to those who are or might become pregnant. Women must produce two negative pregnancy tests and use contraception methods before beginning pomalidomide. Women must commit either to abstain continuously from heterosexual sexual intercourse or to use two methods of reliable birth control, beginning 4 weeks prior to initiating treatment with pomalidomide, during therapy, during dose interruptions and continuing for 4 weeks following discontinuation of pomalidomide therapy. Pomalidomide is present in the semen of patients receiving the drug. Therefore, males must always use a latex or synthetic condom during any sexual contact with females of reproductive potential while taking pomalidomide and for up to 28 days after discontinuing pomalidomide, even if they have undergone a successful vasectomy. Male patients taking pomalidomide must not donate sperm.
