 | ||
Periodate /pəˈraɪ.ədeɪt/ is an anion composed of iodine and oxygen. It is one of a number of oxyanions of iodine and is the highest in the series, with iodine existing in oxidation state +7. Unlike other perhalogenates, such as perchlorate, it can exist in two forms: metaperiodate IO−
4 and orthoperiodate IO5−
6. In this regard it is comparable to the tellurate ion from the adjacent period. It can combine with a number of counter ions to form periodates, which may also be regarded as the salts of periodic acid.
Contents
- Potassium periodate
- Synthesis
- Forms and interconversion
- Structure and bonding
- Cleavage reactions
- Oxidation reactions
- Niche uses
- Other oxyanions
- References
Periodates were discovered by Heinrich Gustav Magnus and C. F. Ammermüller; who first synthesised periodic acid in 1833.
Potassium periodate
Synthesis
Classically, periodate was most commonly produced in the form of sodium hydrogen periodate (Na3H2IO6). This is commercially available, but can also be produced by the oxidation of iodates with chlorine and sodium hydroxide. Or, similarly, from iodides by oxidation with bromine and sodium hydroxide:
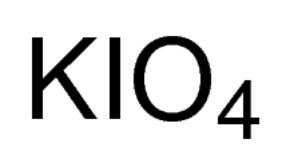
Modern industrial scale production involves the electrochemical oxidation of iodates, on a PbO2 anode, with the following standard electrode potential:
H5IO6 + H+ + 2 e− → IO−3 + 3 H2O E° = 1.6 V
Metaperiodates are typically prepared by the dehydration of sodium hydrogen periodate with nitric acid, or by dehydrating orthoperiodic acid by heating it to 100 °C under vacuum.
Na3H2IO6 + 2 HNO3 → NaIO4 + 2 NaNO3 + 2 H2OH5IO6 → HIO4 + 2 H2OThey can also be generated directly from iodates by treatment with other strong oxidizing agents such as hypochlorites:
NaIO3 + NaOCl → NaIO4 + NaClForms and interconversion
Periodate can exist in a variety of forms in aqueous media, with pH being a controlling factor. Periodate has a number of acid dissociation constants.
H5IO6 ⇌ H4IO−
6 + H+, pKa = 3.29H
4IO−
6 ⇌ H
3IO2−
6 + H+, pKa = 8.31H
3IO2−
6 ⇌ H
2IO3−
6 + H+, pKa = 11.60
The ortho- and metaperiodate forms also exist in equilibrium.
H4IO−
6 ⇌ IO−
4 + 2 H2O, K = 29
For this reason orthoperiodate is sometimes referred to as the dihydrate of metaperiodate, written IO−
4·2H2O; however, this description is not strictly accurate as X-ray crystallography of H5IO6 shows 5 equivalent I–OH groups.
At extremes of pH additional species can form. Under basic conditions a dehydration reaction can take place to form the diperiodate (sometimes referred to as mesoperiodate).
2 H3IO2−
6 ⇌ H
2I
2O4−
10 + 2 H2O, K = 820
Under strongly acid conditions periodic acid can be protonated to give the orthoperiodonium cation.
H6IO+
6 ⇌ H5IO6 + H+, pKa = −0.8
Structure and bonding
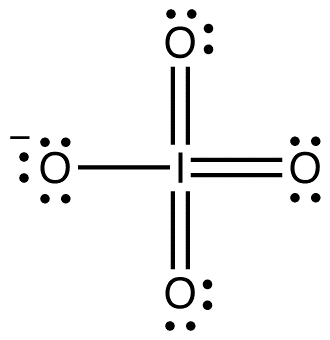
In both the ortho- and metaperiodate the iodine is hypervalent, as it forms more bonds than would classically be allowed. This has been explained in terms of dative bonds, confirming the absence of double bonding in these molecules.
Exact structures vary depending on counter ions, however on average orthoperiodates adopt a slightly deformed octahedral geometry with X-ray diffraction showing I–O bond lengths of 1.89 Å. Metaperiodates adopt a distorted tetrahedral geometry with an average I–O distance of 1.78 Å.
Cleavage reactions
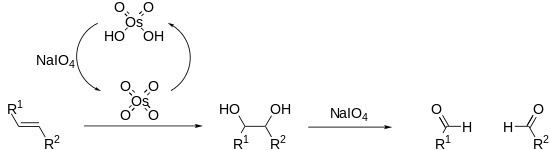
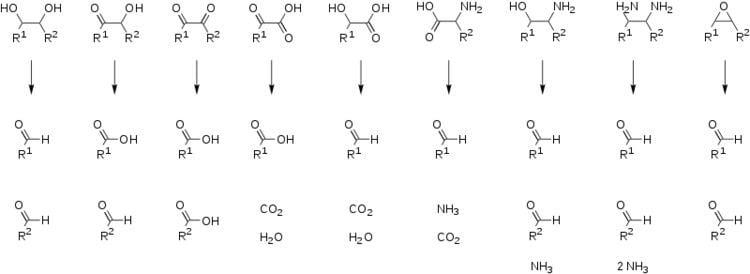
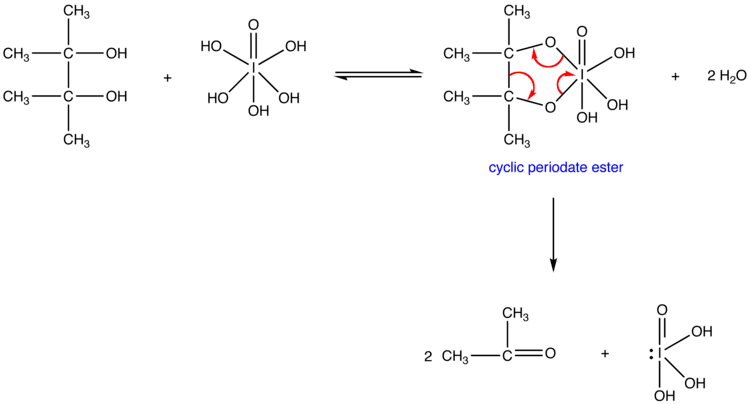
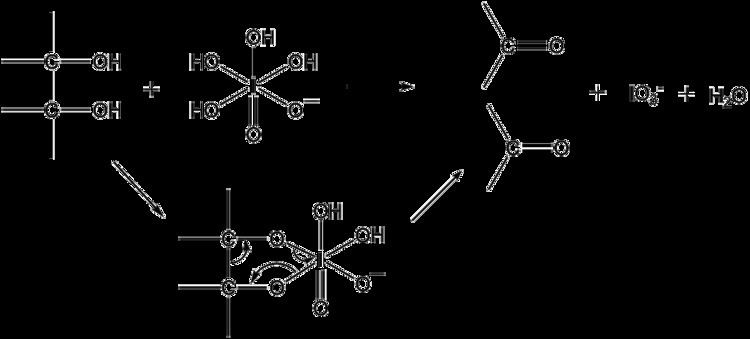
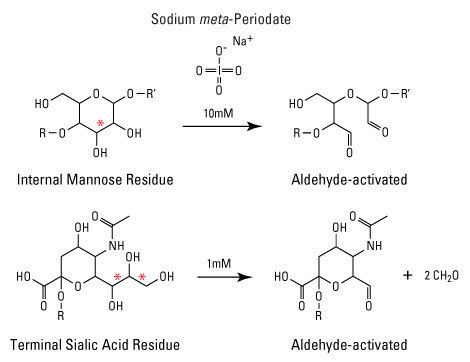
Periodates can cleave carbon–carbon bonds on a variety of 1,2-difunctionalised alkanes. The most common example of this is diol cleavage, which was also the first to be discovered. In addition to diols, periodates can cleave 1,2-hydroxy ketones, 1,2-diketones, α-keto acids, α-hydroxy acids, amino acids, 1,2-amino alcohols, 1,2-diamines, and epoxides to give aldehydes, ketones and carboxylic acids.
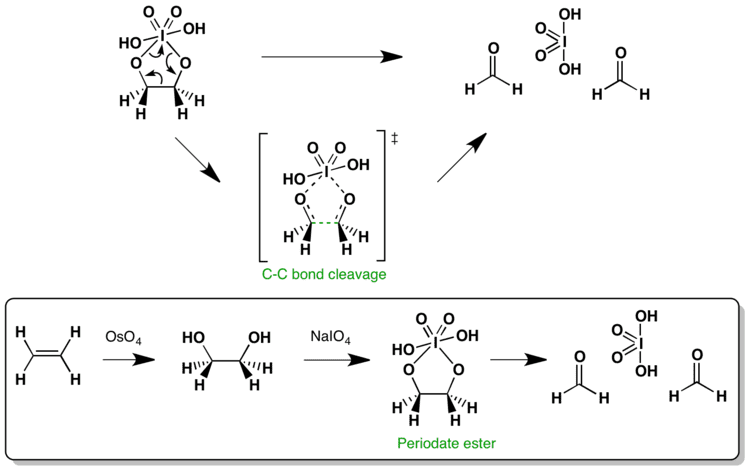
Alkenes can also be oxidised and cleaved in the Lemieux–Johnson oxidation. This uses a catalytic loading of osmium tetroxide which is regenerated in situ by the periodate. The overall process is equivalent to that of ozonolysis.
Reactions proceed via a cyclic intermediate called a periodate ester. The formation of this may be affected by pH and temperature but is most strongly effected by the geometry of the substrate, with cis-diols reacting significantly faster than trans-diols. The reactions are exothermic and are typically performed at 0 °C. As periodate salts are only readily soluble in water reactions are generally performed in aqueous media. Where solubility is an issue periodic acid may be used, as this is soluble in alcohols; phase transfer catalysts are also effective in biphasic reaction mixtures. In extreme cases the periodate may be exchanged for lead tetraacetate which reacts in a similar manner and is soluble in organic solvents.
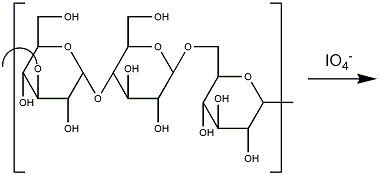
Periodate cleavage is often utilized in molecular biochemistry for the purposes of modifying saccharide rings, as many five- and six-membered sugars have vicinal diols. Historically it was also used to determine the structure of monosaccharides.
Periodate cleavage may be performed on an industrial scale to form dialdehyde starch which has uses in paper production.
Oxidation reactions
Periodates are powerful oxidising agents. They can oxidise catechol to 1,2-benzoquinone and hydroquinone to 1,4-benzoquinone. Sulfides can be effectively oxidised to sulfoxides. Periodates are sufficiently powerful to generate other strong inorganic oxidisers such as permanganate, osmium tetroxide and ruthenium tetroxide.
Niche uses
Periodates are highly selective etchants for certain ruthenium-based oxides.
Several staining agents use in microscopy are based around periodate (e.g. periodic acid–Schiff stain and Jones' stain)
Periodates have also been used as oxidising agents for use in pyrotechnics. In 2013 the US Army announced that it would replace the environmentally harmful chemicals barium nitrate and potassium perchlorate with sodium metaperiodate for use in their tracer ammunition.
Other oxyanions
Periodate is part of a series of oxyanions in which iodine can assume oxidation states of −1, +1, +3, +5, or +7. A number of neutral iodine oxides are also known.
