Related compounds Formula HNO5S Melting point 73.5 °C Pubchem 82157 | Molar mass 127.08 g/mol Density 1.61 g/cm³ Appearance pale yellow crystals | |
 | ||
Attempts to make nitrosylsulfuric acid
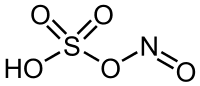
Nitrosylsulfuric acid is the chemical compound with the formula NOHSO4. It is a colourless solid that is used industrially in the production of caprolactam, and was formerly part of the lead chamber process for producing sulfuric acid.
Contents
Synthesis and reactions
A typical procedure entails dissolving sodium nitrite in concentrated sulfuric acid in an ice bath:
HNO2 + H2SO4 → NOHSO4 +H2OThe molecule can also be viewed as the mixed anhydride of sulfuric acid and nitrous acid. Alternatively, it can be prepared by the reaction of nitric acid and sulfur dioxide.
NOHSO4 is useful in organic chemistry to prepare diazonium salts from amines. Related NO-delivery reagents include nitrosonium tetrafluoroborate ([NO]BF4) and nitrosyl chloride.
References
Nitrosylsulfuric acid Wikipedia(Text) CC BY-SA
