Formula CH4N4O2 Boiling point 250 °C Pubchem 11174 | Molar mass 104.07 g/mol Density 1.72 g/cm³ | |
 | ||
Related compounds Appearance Colorless crystalline solid | ||
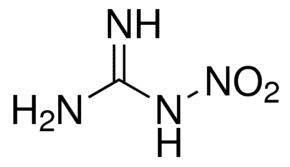
Nitroguanidine is an organic compound with the formula (NH2)2CNNO2. It is a colorless, crystalline solid that melts at 232 °C and decomposes at 250 °C. It is not flammable and is a low-sensitivity explosive; however, its detonation velocity is high. It is used as a propellant (air bags), fertilizer, and for other purposes.
Contents
Manufacture
Nitroguanidine is produced commercially by a two step process starting with the reaction of calcium cyanamide with ammonium nitrate. Via the intermediacy of biguanidine, this ammonolysis step affords the salt guanidinium nitrate. In the second step, the nitrate salt is treated with sulfuric acid, a process that dehydrates the salt and forms the N-N bond.
[C(NH2)3]NO3 → (NH2)2CNNO2 + H2ONitroguanidine can also be generated by treatment of urea with ammonium nitrate. Owing to problems of reliability and safety, this process has not been commerciallized despite its attractive economic features.
Explosives
Nitroguanidine is used as an explosive propellant, notably in triple-base smokeless powder. The nitroguanidine reduces the propellant's flash and flame temperature without sacrificing chamber pressure. These are typically used in large bore guns where barrel erosion and flash are particularly important to avoid.
Nitroguanidine's explosive decomposition is given by the following equation: H4N4CO2 (s) = 2 H2O (g) + 2 N2 (g) + C (s)
Pesticides
Nitroguanidine derivatives are used as insecticides, having a comparable effect to nicotine. Derivatives include clothianidin, dinotefuran, imidacloprid, and thiamethoxam.
Biochemistry
The nitrosoylated derivative nitrosoguanidine is often used to mutagenize bacterial cells for biochemical studies.
Structure
Nitroguanidine exists in two tautomeric forms, as a nitroimine (left) or a nitroamine (right). In solution and in the solid state, the nitroimine form predominates (resonance stabilized).
