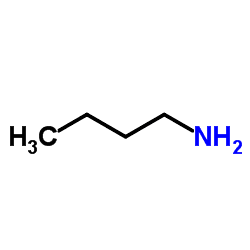
Abbreviations NBA Formula C4H11N | Appearance Colorless liquid Density 740 kg/m³ | |
 | ||
Related alkanamines Related compounds | ||
n-Butylamine is an organic compound (specifically, an amine) with the formula CH3CH2CH2CH2NH2. This colourless liquid is one of the four isomeric amines of butane, the others being sec-butylamine, tert-butylamine and isobutylamine. At standard temperature and pressure, n-butylamine is a liquid having the fishy, ammonia-like odor common to amines. The liquid acquires a yellow color upon storage in air. It is soluble in all organic solvents.
Contents
Like other simple aliphatic amines, n-butylamine is a weak base with a pKa of 10.59 in its protonated form.

Uses

This compound is used as an ingredient in the manufacture of pesticides (such as thiocarbazides), pharmaceuticals, and emulsifiers. It is also a precursor for the manufacture of N,N'-dibutylthiourea, a rubber vulcanization accelerator, and n-butylbenzenesulfonamide, a plasticizer of nylon.
N-Butylamine was used in the synthesis of Fengabine.
Safety
The LD50 to rats through the oral exposure route is 366 mg/kg.
In regards to occupational exposures to n-Butylamine, the Occupational Safety and Health Administration and National Institute for Occupational Safety and Health have set occupational exposure limits at a ceiling of 5 ppm (15 mg/m3) for dermal exposure.
