 | ||
A metal–air electrochemical cell is an electrochemical cell that uses an anode made from pure metal and an external cathode of ambient air, typically with an aqueous electrolyte.
Contents
Types
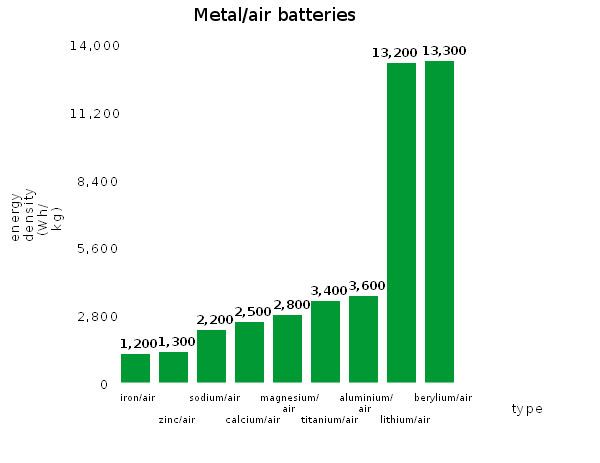
The Li–air battery discharge reaction between Li and oxygen Li2O, according to 4Li + O2 → 2Li2O, has an open-circuit voltage of 2.91 V and a theoretical specific energy of 5,210 watt-hours per kilogram (Wh/kg). Since oxygen is not stored in the battery, the theoretical specific energy excluding oxygen is 11,140 Wh/kg (40.1 MJ/kg). Compare this to the figure of 12,200 Wh/kg (44 MJ/kg) for gasoline (see Petrol energy content).
Iron-air
Iron-air rechargeable batteries are an attractive technology with the potential of grid-scale energy storage. The main raw-material of this technology is iron oxide (rust) which is abundant, non-toxic, inexpensive and environmentally friendly. Most of the batteries being developed right now utilize iron oxide (mostly powders) to generate/store hydrogen via the Fe/FeO reduction/oxidation (redox) reaction (Fe + H2O = FeO + H2). In conjunction with a fuel cell this enables the system to behave as a rechargeable battery creating H2O/H2 via production/consumption of electricity. Furthermore, this technology has minimal environmental impact as it could be used to store energy from intermittent solar and wind power sources, developing an energy system with low carbon dioxide emissions.
The way the system works can start by using the Fe/FeO redox reaction, then the hydrogen created during the oxidation of iron can be consumed by a fuel cell in conjunction with oxygen from the air to create electricity. When electricity must be stored, hydrogen generated from water by operating the fuel cell in reverse is consumed during the reduction of the iron oxide to metallic iron. The combination of both of this cycles is what makes the system operate as an iron-air rechargeable battery.
Limitations of this technology come from the materials used. Generally, iron oxide powder beds are selected, however, rapid sintering and pulverization of the powders limit the ability to achieve a high number of cycles resulting in a lower capacity. Other methods, such as 3D-Printing and freeze casting, currently under investigation seek to enable the creation of architecture materials to allow for high surface area and volume changes during the redox reaction.
