 | ||
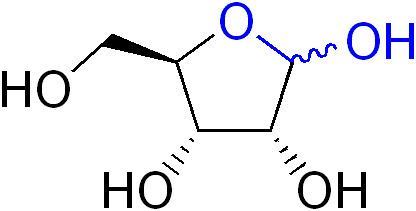
In organic chemistry, a lactol is the cyclic equivalent of a hemiacetal or a hemiketal. The compound is formed by the intramolecular nucleophilic addition of a hydroxyl group to the carbonyl group of an aldehyde or a ketone.
Contents
A lactol is often found as an equilibrium mixture with the corresponding hydroxyaldehyde. The equilibrium can favor either direction depending on ring size and other conformational effects.
The lactol functional group is prevalent in nature as component of aldose sugars.
Beaphar lactol kitty milk
Chemical reactivity
Lactols can participate in a variety of chemical reactions including:
References
Lactol Wikipedia(Text) CC BY-SA
