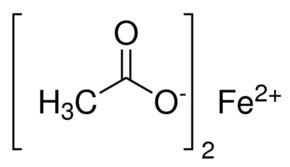
Formula Fe(C2H3O2)2 | Molar mass 173.93 g/mol | |
 | ||
Appearance White crystals (anhydrous); Light green crystals (tetrahydrate) | ||
Iron(II) acetate is an coordination complex with formula Fe(C2H3O2)2. It is a white solid, although impure samples can be slightly colored. A light green tetrahydrate is also known, which is highly soluble in water.
Contents
Preparation and structure
Iron powder reacts with hot acetic acid to give the product:
Fe + 2 CH3CO2H → Fe(CH3CO2)2 + H2It adopts a polymeric structure with octahedral Fe(II) centers bridged by acetate ligands. It is not a salt.
The hydrate can be made by the reaction of ferrous oxide or ferrous hydroxide with acetic acid.
Reaction of scrap iron with acetic acid affords a brown mixture of various iron(II) and iron(III) acetates that are used in dyeing.
Uses
Ferrous acetate is used as a mordant by the dye industry. Ebonizing wood is one such process.
References
Iron(II) acetate Wikipedia(Text) CC BY-SA
