Formula I2Cl6 Melting point 63 °C Appearance yellow solid | Molar mass 466.5281 g/mol Density 3.11 g/cm³ | |
 | ||
Vsepr theory iodine trichloride icl3 expanded valence
Iodine trichloride is an interhalogen compound of iodine and chlorine. It is bright yellow and in the solid state is present as a planar dimer I2Cl6, Cl2I(μ-Cl)2ICl2, with two bridging Cl atoms.
Contents

It can be prepared by reacting iodine with an excess of liquid chlorine at −70 °C. In the melt it is conductive, which may indicate dissociation:
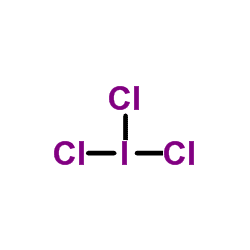
Iodine trichloride can be created by heating a mixture of liquid iodine and chlorine gas to 105 °C.
It is an oxidizing agent, capable of causing fire on contact with organic materials.
Vsepr theory iodine trichloride icl3 expanded valence
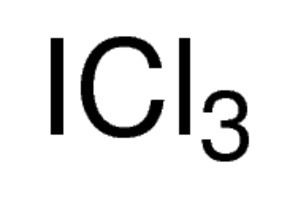
References
Iodine trichloride Wikipedia(Text) CC BY-SA
