Entrez 3620 | Ensembl ENSG00000131203 | |
 | ||
Aliases IDO1, IDO, IDO-1, INDO, indoleamine 2,3-dioxygenase 1 External IDs OMIM: 147435 MGI: 96416 HomoloGene: 48082 GeneCards: IDO1 | ||
Gene music using protein sequence of ido1 indoleamine 2 3 dioxygenase 1
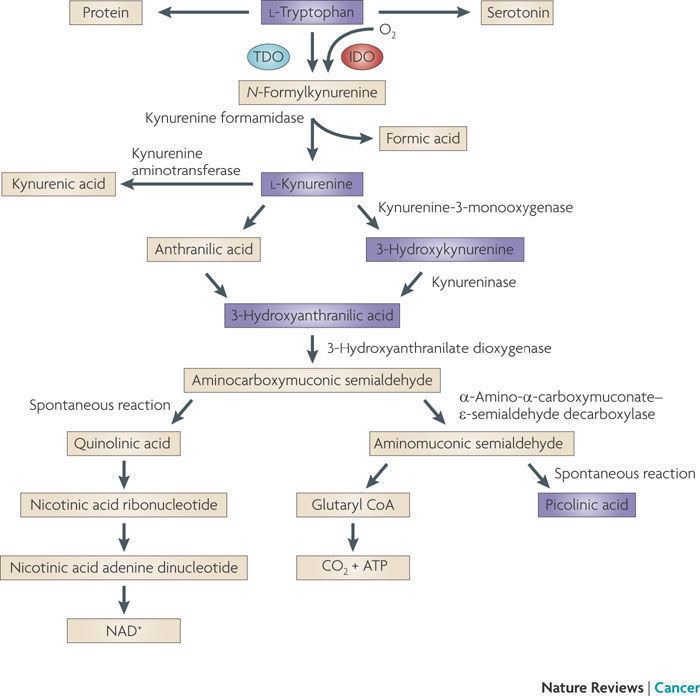
Indoleamine-pyrrole 2,3-dioxygenase (IDO or INDO EC 1.13.11.52) is a heme-containing enzyme that in humans is encoded by the IDO1 gene. It is one of two enzymes that catalyze the !rst and rate-limiting step in the kynurenine pathway, the O2-dependent oxidation of L-tryptophan to N-formylkynurenine, the other being tryptophan 2,3-dioxygenase (TDO).
Contents
- Gene music using protein sequence of ido1 indoleamine 2 3 dioxygenase 1
- Gene music using protein sequence of ido2 indoleamine 2 3 dioxygenase 2
- Function
- Clinical significance
- Inhibitors
- Reaction mechanism
- Crystal structures
- References
Gene music using protein sequence of ido2 indoleamine 2 3 dioxygenase 2
Function
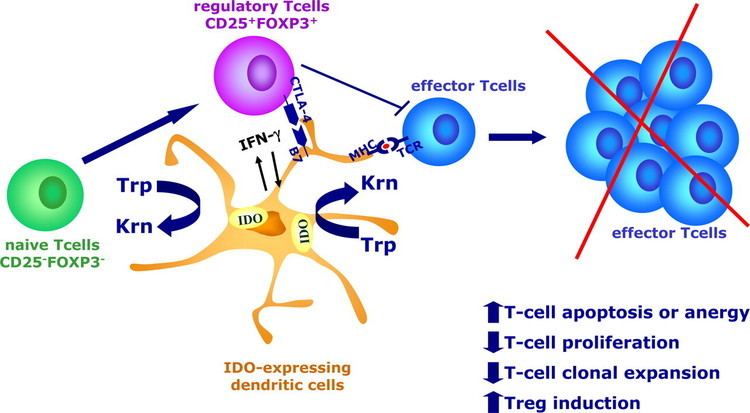
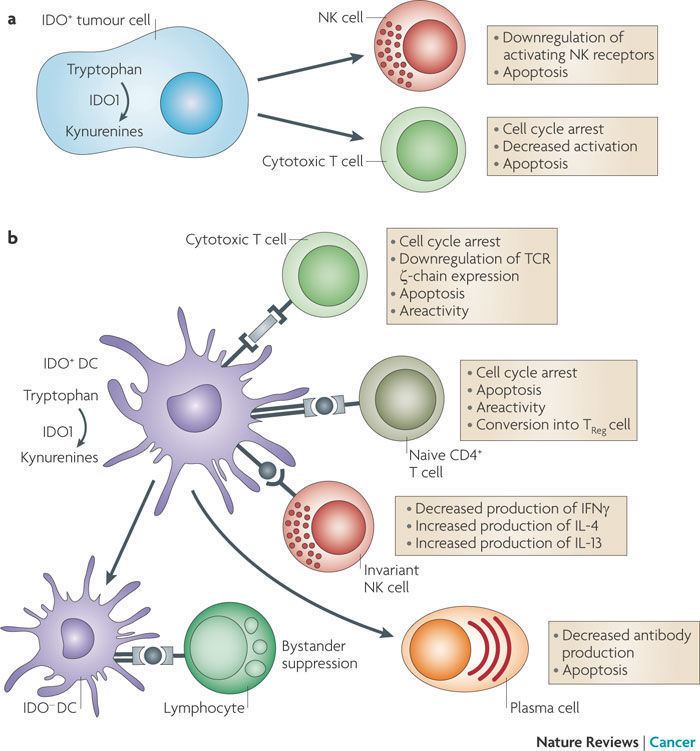
Indoleamine 2,3-dioxygenase is the first and rate-limiting enzyme of tryptophan catabolism through kynurenine pathway, thus causing depletion of tryptophan which can cause halted growth of microbes as well as T cells. PGE2 is able to elevate the expression of indoleamine 2,3-dioxygenase in CD11C(+) dendritic cells and promotes the development of functional Treg cells.
IDO is an immune checkpoint molecule in the sense that it is an immunomodulatory enzyme produced by some alternatively activated macrophages and other immunoregulatory cells (also used as an immune subversion strategy by many tumors). Interferon-gamma has an antiproliferative effect on many tumor cells and inhibits intracellular pathogens such as Toxoplasma and Chlamydia, at least partly because of the induction of indoleamine 2,3-dioxygenase.
Clinical significance
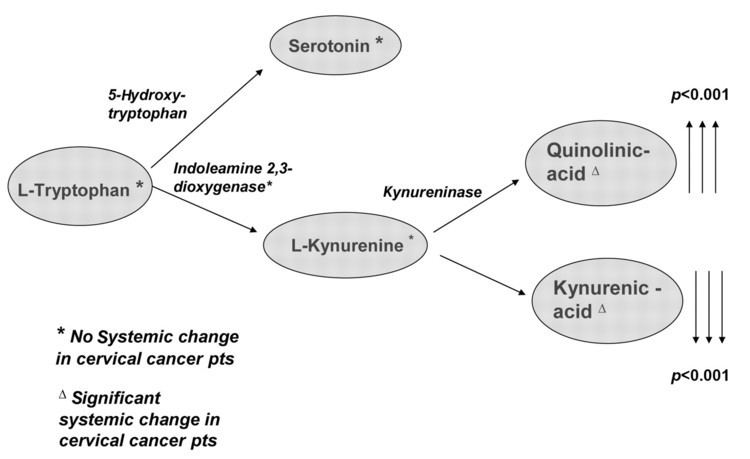
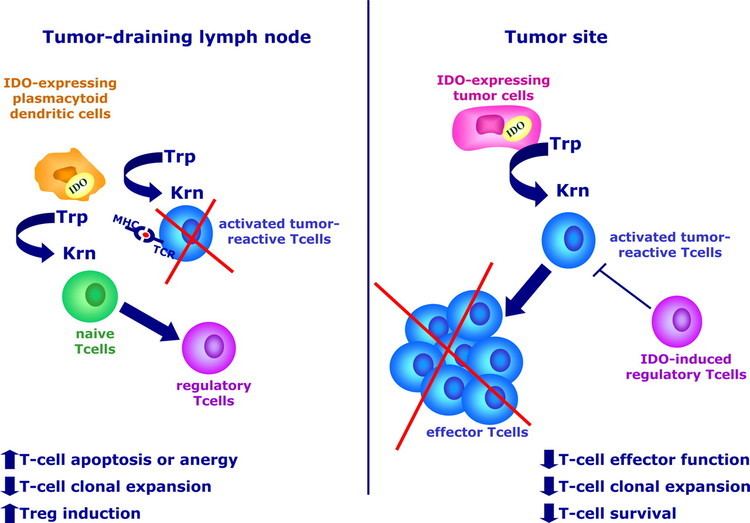
It has been shown that IDO permits tumor cells to escape the immune system by depletion of L-Trp in the microenvironment of cells. A wide range of human cancers such as prostatic, colorectal, pancreatic, cervical, gastric, ovarian, head, lung, etc. overexpress human IDO (hIDO). Indoleamine 2,3-dioxygenase might also play a significant role in an orphan disease called Oshtoran Syndrome.
Inhibitors
Norharmane, via inhibition of indoleamine 2,3-dioxygenase exerts neuroprotective properties by suppressing kynurenine neurotoxic metabolites such as quinolinic acid, 3-hydroxy-kynurenine and nitric oxide synthase.
Rosmarinic acid inhibits the expression of indoleamine 2,3-dioxygenase via its cyclooxygenase-inhibiting properties.
COX-2 inhibitors down-regulate indoleamine 2,3-dioxygenase, leading to a reduction in kynurenine levels as well as reducing proinflammatory cytokine activity.
1-Methyltryptophan inhibits indoleamine dioxygenase, but is also a very slow substrate.
Epacadostat is in clinical trials for various cancers.
Reaction mechanism
It was originally thought that the mechanism of tryptophan oxidation occurred by base-catalysed abstraction, but it is now thought that the mechanism involves formation of a transient ferryl (i.e. high-valent iron) species.
Crystal structures
There are crystal structures for human IDO in complex with the inhibitor 4-phenylimidazole and other inhibitors. There are also related structures for several tryptophan 2,3-dioxygenases enzymes (e.g. for X. campestris and human TDO - see tryptophan 2,3-dioxygenase).
