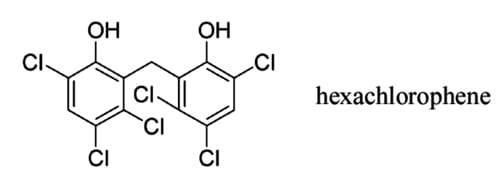
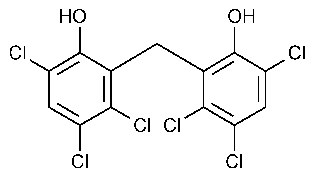
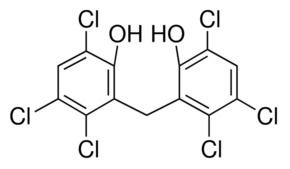
CAS Number 70-30-4 DrugBank DB00756 CAS ID 70-30-4 | PubChem CID 3598 Formula C13H6Cl6O2 Density 1.71 g/cm³ | |
 | ||
Trade names pHisoHex, Gamophen, Septisol, Turgex, Germa-Medica, Hexachlorophane, Almederm ATC code D08AE01 (WHO) QP52AG02 (WHO) Legal status US: ℞-only for human useRx-only for human use | ||
Medical vocabulary what does hexachlorophene mean
Hexachlorophene, also known as Nabac, is an organochlorine compound that was once widely used as a disinfectant. The compound occurs as a white odorless solid, although commercial samples can be off-white and possess a slightly phenolic odor. It is insoluble in water but dissolves in acetone, ethanol, diethyl ether, and chloroform. In medicine, hexachlorophene is a useful as a topical anti-infective, anti-bacterial agent, often used in soaps and toothpaste. It is also used in agriculture as a soil fungicide, plant bactericide, and acaricide.
Contents
- Medical vocabulary what does hexachlorophene mean
- US and removal from market
- Europe
- Production
- Safety
- Trade names
- References
US, and removal from market

In 1972, the U.S. Food and Drug Administration (FDA) halted the production and distribution of products containing more than 1% of hexachlorophene. After that point, most products that contain hexachlorophene were available only by prescription from a doctor. The restrictions were enacted after 15 deaths in the United States and 39 deaths in France were reported following brain damage caused by hexachlorophene.
At least two companies manufactured over-the-counter preparations incorporating hexachlorophene. One product was Baby Magic Bath by The Mennen Company. Mennen recalled the product in 1971, and it was removed from retail distribution. Immediately after the withdrawal, there was an outbreak of Staphylococcus infections in hospitals across the USA.
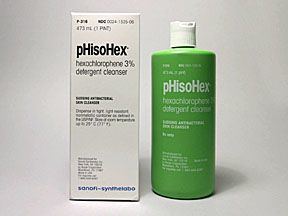
Two commercial preparations using hexachlorophene, pHisoDerm and pHisoHex, were widely used as antibacterial skin cleansers in the treatment of acne, with pHisoDerm developed for those allergic to the active ingredients in pHisoHex. In the US during the 1960s, both were available over the counter. After the ban was enacted, pHisoDerm was reformulated without hexachlorophene, and continued to be sold over-the-counter, while pHisoHex, which contained 3% hexachlorophene (three times the legal limit imposed in 1972), became available (and remains available today) as a prescription body wash. In the European Community countries during the 1970s and 1980s, pHisoHex was available over the counter. A related product, pHisoAc, was used as a skin mask to dry and peel away acne lesions. Another preparation, pHiso-Scrub, was a hexachlorophene-impregnated sponge for scrubbing, has since been discontinued. Several substitute products (including triclosan) were developed, but none had the germ-killing capability of hexachlorophene. Sanofi-Aventis was the sole manufacturer of pHisoHex, while The Mentholatum Company owns the pHisoDerm brand today. Sanofi-Aventis discontinued production of several forms of pHisoHex in August 2009 and discontinued all production of pHisoHex in September 2013.
The formula for Dial soap was changed to remove hexachlorophene after the FDA put an end to over-the-counter availability in 1972.
Europe
In Germany, cosmetics containing hexachlorophene have been forbidden since 1985. In Austria, sale of drugs containing the substance has been forbidden since 1990.
Production
Hexacholorophene is produced by alkylation of 2,4,5-trichlorophenol with formaldehyde. Related antiseptics are prepared similarly, e.g., bromochlorophene and dichlorophene.
Safety
The LD50 (oral, rat) is 59 mg/kg, indicating that the compound is relatively toxic. It is not mutagenic nor teratogenic according to Ullmann's Encyclopaedia, but "embryotoxic and produces some teratogenic effects" according to the International Agency for Research on Cancer.
Trade names
Trade names for hexachlorophene include: Acigena, Almederm, AT7, AT17, Bilevon, Exofene, Fostril, Gamophen, G-11, Germa-Medica, Hexosan, K-34, Septisol, Surofene.
