MedlinePlus a684001 Routes ofadministration Oral Molar mass 352.7 g/mol | Pregnancycategory ? ATC code N05BA13 (WHO) CAS ID 23092-17-3 | |
 | ||
AHFS/Drugs.com Micromedex Detailed Consumer Information Legal status CA: Schedule IVDE: Anlage III (Prescription only)US: Schedule IV | ||
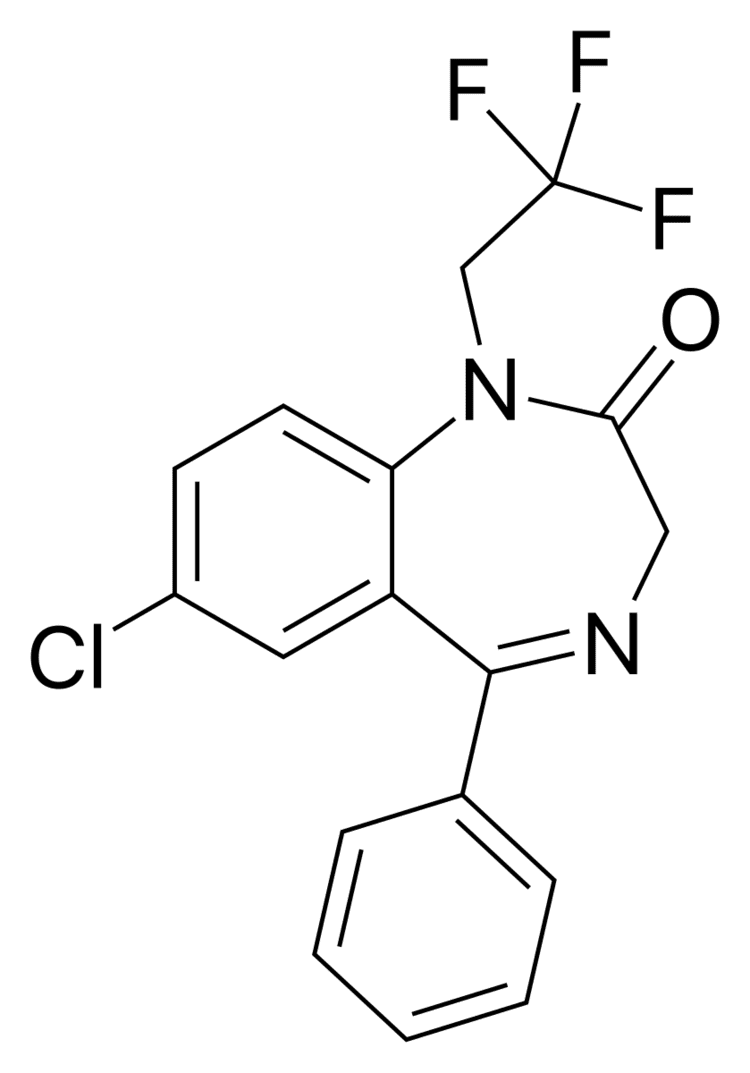
Halazepam is a benzodiazepine derivative that was marketed under the brand names Paxipam in the United States, Alapryl in Spain, and Pacinone in Portugal.
Contents
- What does halazepam mean
- Medical uses
- Adverse effects
- Pharmacokinetics and pharmacodynamics
- Regulatory Information
- Commercial production
- References
What does halazepam mean
Medical uses
Halazepam was used for the treatment of anxiety.
Adverse effects
Adverse effects include drowsiness, confusion, dizziness, and sedation. Gastrointestinal side effects have also been reported including dry mouth and nausea.
Pharmacokinetics and pharmacodynamics
Pharmacokinetics and pharmacodynamics were listed in Current Psychotherapeutic Drugs published in June 15, 1998 as follows:
Regulatory Information
Halazepam is classified as a schedule 4 controlled substance with a corresponding code 2762 by the Drug Enforcement Administration (DEA).
Commercial production
Halazepam was invented by Schlesinger Walter in the U.S. It was marketed as an anti-anxiety agent in 1981. However, Halazepam is not commercially available in the United States because it was withdrawn by its manufacturer for poor sales.
