 | ||
The group 2 elements are known to form organometallic compounds. Of these, organomagnesium compounds, usually in the form of Grignard reagents are widely used in organic chemistry, while the other organometallic compounds of this group are largely academic.
Contents
Characteristics
In many ways the chemistry of group 2 elements (the alkaline earth metals) mimics that of group 12 elements because both groups have filled s shells for valence electrons. Thus, both groups have nominal valency 2 and oxidation state +2. All group 2 elements are electropositive towards carbon and electronegativity decreases down the row. At the same time the atomic radius increases resulting in increasingly ionic character, higher coordination numbers, and increased ligand reactivity.
Many dialkyl group 2 metals are polymeric in the crystalline phase and resemble trimethylaluminium in three-center two-electron bond. In the gas-phase they are once again monomeric.
The metallocenes in this group are unusual. Bis(cyclopentadienyl)beryllium or beryllocene (Cp2Be) with a molecular dipole moment of 2.2 D rules out a classical metallocene with two hapticity 5 ligands. Instead the compound is a so-called slip 5η/1η sandwich and on top of that also fluxional up to −125 °C. While magnesocene (Cp2Mg) is a regular metallocene, bis(pentamethylcyclopentadienyl)calcium (Cp*)2Ca is actually bent with an angle of 147°. This angle increases going down the row.
Low-valent organometallics with formal oxidation state 1 having a metal to metal bond are also known. A representative is LMg-MgL with L = [(Ar)NC(NPri2)N(Ar)]−.
Synthesis
Three important ways for synthesis of dialkyl and diaryl group 2 metal compounds is by metathesis:
MX2 + R-Y → MR2 + Y-X'By transmetallation:
M'R2 + M → MR2 + M'Manipulation of the Schlenk equilibrium of the organometal halides:
2 RMX → MR2 + MX2See for example the formation of dimethylmagnesium.
Organoberyllium
Organoberyllium chemistry is limited to academic research due to the cost and toxicity of beryllium, beryllium derivatives and reagents required for the introduction of beryllium, such as beryllium chloride. Organometallic beryllium compounds are known to be highly reactive Examples of known organoberyllium compounds are dineopentylberyllium, beryllocene (Cp2Be), diallylberyllium (by exchange reaction of diethyl beryllium with triallyl boron), bis(1,3-trimethylsilylallyl)beryllium and Be(mes)2. Ligands can also be aryls and alkynyls.
Organomagnesium
Organomagnesium compounds are widespread. They are commonly found as Grignard reagents. The formation of alkyl or aryl magnesium halides (RMgX) from magnesium metal and an alkyl halide is attributed to a SET process. Examples of Grignards are phenylmagnesium bromide and ethylmagnesium bromide.
Relevant organic magnesium reagents outside the scope of Grignards are magnesium anthracene with magnesium forming a 1,4-bridge over the central hexagon used as a source of highly active magnesium and butadiene magnesium an adduct with butadiene and a source for the butadiene dianion.
Organocalcium
Further down this group calcium is nontoxic and cheap but organocalcium compounds are difficult to make. This is even more so for the remaining members strontium and barium, and for the case of radium there are none known at all. One use for this type of compounds is in chemical vapor deposition.
A well known organocalcium compound is (Cp)calcium(I). Bis(allyl)calcium was described in 2009. It forms in a metathesis reaction of allylpotassium and calcium iodide as a stable non-pyrophoric off-white powder:
The bonding mode is η3. This compound is also reported to give access to an η1 polymeric (CaCH2CHCH2)n compound.
The compound [(thf)3Ca{μ-C6H3-1,3,5-Ph3}Ca(thf)3] also described in 2009 is an inverse sandwich compound with two calcium atoms at either side of an arene.
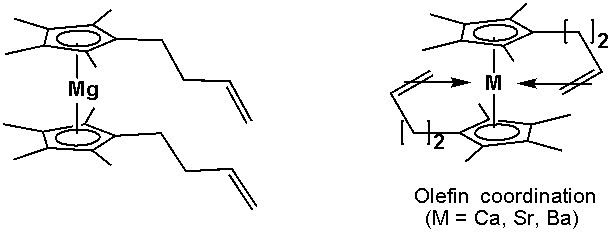
Olefins tethered to cyclopentadienyl ligands have been shown to coordinate to calcium(II), strontium(II), and barium(II):
Organocalcium compounds are investigated as catalysts.
Organostrontium
Organostrontium compounds have been reported as intermediates in Barbier-type reactions.
Organobarium
Organobarium compounds of the type (allyl)BaCl are known and can be prepared by reaction of activated barium (Rieke method reduction of barium iodide with lithium biphenylide) with allyl halides at −78 °C. Subsequent reaction of these allylbarium compounds with carbonyl compounds is reported to be more alpha-selective and more stereoselective than the related Grignards or organocalcium compounds. The metallocene (Cp*)2Ba has also been reported.
Organoradium
The only known organoradium compound is the gas-phase acetylide.
