Symbol Glyco_hydro_38 Pfam clan CL0158 SCOP 1o7d | Pfam PF01074 InterPro IPR000602 SUPERFAMILY 1o7d | |
 | ||
In molecular biology, glycoside hydrolase family 38 is a family of glycoside hydrolases.
Glycoside hydrolases EC 3.2.1. are a widespread group of enzymes that hydrolyse the glycosidic bond between two or more carbohydrates, or between a carbohydrate and a non-carbohydrate moiety. A classification system for glycoside hydrolases, based on sequence similarity, has led to the definition of >100 different families. This classification is available on the CAZy(http://www.cazy.org/GH1.html) web site, and also discussed at CAZypedia, an online encyclopedia of carbohydrate active enzymes.
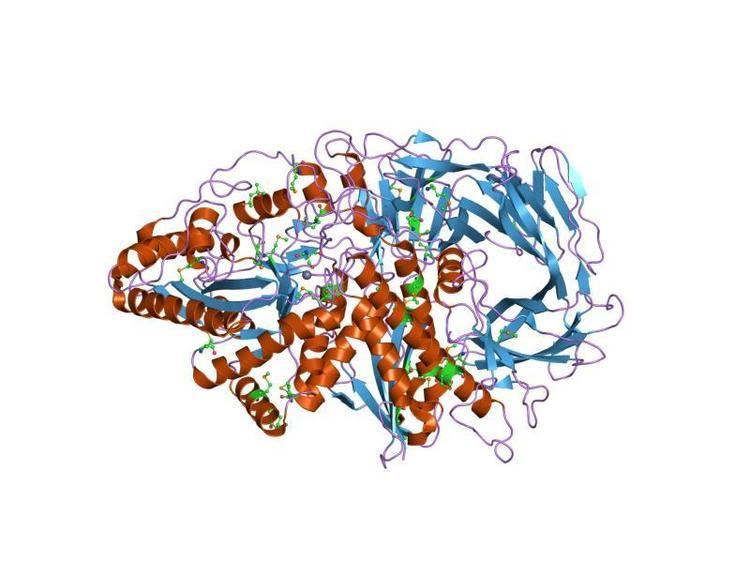
Glycoside hydrolase family 38 CAZY GH_38 comprises enzymes with only one known activity; alpha-mannosidase (EC 3.2.1.24) (EC 3.2.1.114).
Lysosomal alpha-mannosidase is necessary for the catabolism of N-linked carbohydrates released during glycoprotein turnover. The enzyme catalyzes the hydrolysis of terminal, non-reducing alpha-D-mannose residues in alpha-D-mannosides, and can cleave all known types of alpha-mannosidic linkages. Defects in the gene cause lysosomal alpha-mannosidosis (AM), a lysosomal storage disease characterised by the accumulation of unbranched oligo-saccharide chains.
A domain, which is found in the central region adopts a structure consisting of three alpha helices, in an immunoglobulin/albumin-binding domain-like fold. The domain is predominantly found in the enzyme alpha-mannosidase.
